Assignment 1, Part 4: Measuring magnification and bead size
Measure the microscope's magnification
Measuring the magnification of your microscope is a good way to verify that your instrument has been constructed correctly. (In general, you should measure the magnification of any microscope you will use to make quantitative measurements of size). Use the measured value in your calculations, not the number printed on the objective.
- Install the 10x objective in your microscope and image the microruler calibration slide.
- You will find the objectives in the west drawers of the lab. Be sure to pick up a RMS to SM1 adaptor from the neighboring bin to be able to thread the objective into the Thorlabs cage cube.
- The microruler calibration slide has tick marks that are 10 μm apart. Every 100 μm, there is a longer tick mark.
- Make sure that the side of the microruler with the pattern on it faces the objective. Imaging through the thick glass causes distortion and many other troubles.
- Start the live preview using the UsefulImageAcquisition tool:
- Familiarize yourself with the UsefulImageAcquisition tool and how to save and display images by reading through these instructions.
- Ensure that the camera's field of view is approximately centered in the objective's field of view.
- The objective has a larger field of view (FOV) than the camera. Adjust the 45 degree mirror to traverse the objective's FOV horizontally and vertically. The FOV is approximately circular. Find a spot near the middle.
- Record an image of the microruler.
- Once your happy with the image on the live preview, click the Acquire button.
- Display the image and use imdistline or the data cursor to measure a known distance between rulings in your image and compute the magnification.
- When choosing a distance to measure, consider the factors that influence the uncertainty of your measurement.
- Save your images in a .mat file for later use in MATLAB or as a PNG image for use in your report or other programs (Did you read the instructions??).
- Repeat the magnification measurement for the 40x and 100x objectives.
- Using your magnification measurements and the known size of the CMOS camera, calculate the field of view (FOV) of the microscope for each objective.
| Manufacturer specified magnification | As-designed magnification | Object height (μm) | Image height (μm) | Measured magnification | Field of view (FOV, μm x μm) |
| 10x | |||||
| 40x | |||||
| 100x |
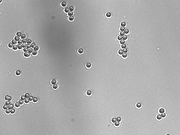
Measure particle size
Now that you know the magnification of your instrument, use it to measure the size of some microscopic objects as imaged with the 40x objective lens only. Slides with 7.2 μm and 3.2 μm silica microspheres are available in the lab.
- Image 7.2 μm and 3.2 μm silica microspheres using the 40x objective only.
- Measure the size of several beads of each type.
- Try to measure beads that are relatively sparse or not too clumped together.
How many spheres should you measure?
- One typical way to estimate the uncertainty in a measurement is to make many measurements of the same thing, and then calculate the standard error. Standard error is equal to the sample standard deviation divided by the square root of the number of data points. It is an estimate of how well you know the mean value of whatever you are measuring. Assuming nothing is changing with your measurement over time, increasing the number of data points will reduce your standard error. On the flip side, you cannot sit in lab forever and make an infinite number of measurements to reduce your uncertainty to zero. What do you think should determine how many measurements you take?
Reporting uncertainty:
- All measured values should be reported with an associated measure of variability, which is usually the range, standard deviation, or standard error of the dataset. Use the abbreviation "s.d." for standard deviation and "s.e.m." for standard error after the "±". For example: 1.21 ± 0.03 GW (±s.d., N=42). Uncertainty is typically reported with one or two significant figures. Round uncertain quantities to the same decimal place as the uncertainty. The sample size must be included in all cases. Report uncertainty in the same units as the measurand. [1] Standard error is typically the best choice for datasets that contain 20 or more samples. It can be interpreted as an estimate of the size of an interval that would contain the result of repeating the experiment about ⅔ of the time. Range or standard deviation is a good choice for small values of N.
Microscope storage
During the microscopy lab, approximately seven thousand optical components will be taken from stock, assembled into microscopes, and properly returned to their assigned places. Please observe the following:
- Store your microscope in one of the cubby holes in 16-336 (not in the lab). If you use one of the high shelves, get somebody to help you lift.
- Keep all of the boxes for the optics you use with your instrument to simplify putting things away.
- Take a blue bin to store loose items (such as lens boxes) in.
- Stages, CCD cameras, neutral density filters and emission filters stay at the lab station. Do not store these with your microscope.
- Return objective lenses to the drawer when you are not using them. (Do not store them with your microscope.)
- The stages are very expensive. Always lift from the bottom.
- If you break something (or discover something pre-broken for you), do not return it to the component stock. Give all broken items to an instructor. You will not be penalized for breaking something, but not reporting may be looked upon less kindly.
References
- ↑ Relative (percent) uncertainty is undesirable in the presence of additive noise because a constant magnitude error source produces different error values as the measurand changes. Relative error is a more sensible choice when measuring an unvarying quantity such as a physical constant.
- Overview
- Part 1: Pre-lab questions
- Part 2: Optics bootcamp
- Part 3: Build a microscope
- Part 4: Measure stuff
Back to 20.309 Main Page