20.109(F20):M1D4
Contents
Introduction
As a brief reminder, two antibodies were used in the H2AX assay. The first antibody, or primary antibody, was anti-γH2AX and raised in a mouse. The secondary antibody was anti-mouse and raised in a goat, more importantly, this molecule is conjugated to a fluorescent dye tag called Alexa Fluor 488. The Alexa Fluor 488 tag is a bright, green fluorescent dye that is excited at 488 nm. To visualize the abundance of double-strand breaks in your H2AX assay samples, we will use fluorescence microscopy.
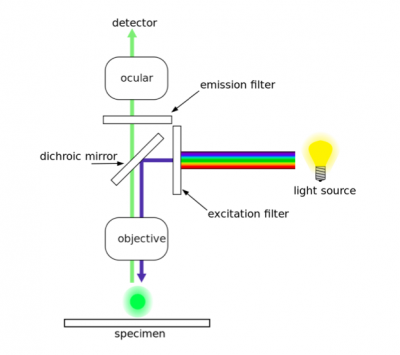
In fluorescence microscopy the specimen is illuminated with a wavelength of light specific to the excitation of the fluorescent tag used to target the feature of interest. The excitation wavelength is absorbed by the fluorescent tag, which causes it to emit light at a longer, less energetic wavelength. Typically, fluorescence microscopes used in biology are an epifluorescence type with a single light path (the objective) for excitation and emission detection, as depicted in the diagram above.
Fluorescence, or epifluorescence, microscopes are composed of a light source, an excitation filter, a dichroic mirror, and an emission filter. The filters and the dichroic mirror are specific to the spectral excitation and emission characteristics of the fluorescent tag. To visualize fluorescence, light at the excitation wavelength is focused on the sample. The light emission from the sample is focused by the objective to a detector.
Protocols
Part 1: Participate in Comm Lab workshop
Our communication instructors, Dr. Prerna Bhargava and Dr. Sean Clarke, will join us today for a discussion on writing Titles and Abstracts.
Part 2: Finish staining procedure for γH2AX assay
To ensure the steps required for preparing the γH2AX experiment for imaging are clear, the Instructor will provide a live demonstration of this process.
In your laboratory notebook, complete the following:
- Provide a written overview / description of the the procedure used to prepare the γH2AX experiment for imaging (from the live demonstration).
- Why is it important to wash the secondary antibody from the coverslip before imaging?
- What stain is included in the mounting media? What cellular component is stained in this step? And why is this useful?
Part 3: Image γH2AX experiment
To ensure you are familiar with the steps involved in imaging the γH2AX experiment, please watch the video tutorial linked here: [H2AX Imaging].
Part 4: Analyze γH2AX images by measuring fluorescence intensity
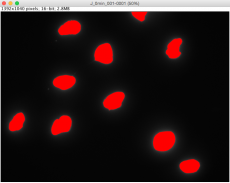
Please obtain the raw γH2AX images from the Class Dropbox folder. Note that there are two different experiments as indicated by date. Six sets of images (i.e. image stacks) were taken per experimental condition, and each image stack contains images from two channels: DAPI (blue) and FITC (green). Remember that the secondary antibody used for the γH2AX staining was conjugated to an Alexa488 fluorophore, which emits green light. For each image stack, you will use ImageJ to 1) identify the location of the nuclei using the DAPI channel and 2) quantify the total γH2AX fluorescence in the FITC channel at locations specified by the DAPI channel.
Identify intensity thresholds for DAPI channel
First, you will identify intensity thresholds that will properly identify the cell nuclei in all the images. To be consistent and fair in analyzing fluorescence images, it is good practice to use the same intensity thresholds on all the images.
- Open ImageJ.
- Open one image stack from the no treatment condition.
- The first image you see is the DAPI channel
- If you scroll to the right, the second image in the stack is the FITC (γH2AX) channel.
- While the image is on the DAPI channel, go to Image -> Adjust -> Threshold.
- A threshold window should pop up
- Check the box for "Dark Background"
- Make sure the cell nuclei are highlighted in red.
- Adjust the threshold values to properly identify the majority of the cells' nuclei.
- Record the threshold values.
- Repeat this process for one image from each condition and cell line, and settle on threshold values for the DAPI channel that you will then use to analyze all the images. Write these values in your notebook.
- It is best to define the lower threshold value based on your images, and set the upper threshold value as 256, which is the maximum possible intensity value for a 8-bit image.
- You can type in threshold values by clicking on the "Set" button in the Threshold window.
- Close all open images (File -> Close All).
Test γH2AX quantification on one representative image
- In ImageJ, open one image to test the FITC quantification protocol.
- Split the image stack into two separate images.
- Go to Image -> Stacks -> Stack to Images.
- The DAPI image will have "-0001" as a suffix in its title.
- The FITC (gamma-H2AX) image will have "-0002" as a suffix in its title.
- Duplicate the DAPI image and turn it into a mask to identify nuclei locations.
- Click on the DAPI image.
- Go to Image -> Duplicate, and click OK on the default title.
- Set the thresholds you chose on the duplicated DAPI image to identify nuclei.
- Go to Image -> Adjust -> Threshold.
- Check the box for "Dark Background".
- Click on the "Set" button and type in your threshold values (use 256 for the upper threshold level).
- Go to Process -> Binary -> Convert to Mask.
- This makes the image black and white, where the white areas should correspond to nuclei locations.
- Use the newly created mask to identify locations on the Texas Red channel in which to quantify the gamma-H2AX signal.
- Go to Analyze -> Set Measurements.
- In the Set Measurements window, make sure the following boxes are checked: Area, Mean gray value, Min & max gray value, Shape descriptors, Integrated density, Display label.
- In the "Redirect to" field, scroll and select the FITC image (suffix -0002). Then press OK.
- This will direct ImageJ to the FITC image to analyze the metrics you selected in the areas identified by your mask. This will give you information about the gamma-H2AX signal in each nucleus.
- Go to Analyze -> Set Measurements.
- Run the analysis by selecting Analyze -> Analyze Particles.
- In the "Size" field, type 200-Infinity. This will eliminate small, extraneous particles that do not correspond to nuclei.
- "Circularity" can remain at default values: 0-1.
- "Show" should say "Outlines".
- Click the following options: Display results, Exclude on edges, Summarize.
- Press OK to complete.
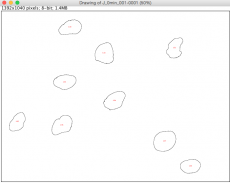
- A window will pop up showing outlines of each nucleus the software identified based on the thresholds you defined. Each identified area is labeled with a red number, corresponding to the left column of the data shown in the "Results" window.
- Take a look at the "Results" window to see the results of the analysis. It is good practice to validate the numerical results by comparing them to what you see in the images.
- The definition of the various measurements performed can be found on the ImageJ website (linked here).
- Does the nucleus with the largest "Area" correspond to the biggest nucleus you see in the drawing? The area here is in units of square pixels.
- The RawIntDens field is the total intensity (sum of the intensity of all the pixels) of the corresponding region. Does a region with a high total intensity value correspond to a cell with a high gamma-H2AX signal? Click on the FITC image to double check.
- Close the "Results" window and do not save the data, as you will run the analysis on all the files together next.
- Close all open windows in ImageJ (File -> Close All).
Quantify γH2AX signal in all images
- Create four folders on your desktop for each experimental condition: No treatment, 100uM H2O2, 2uM Arsenic and 2uM Arsenic+100uM H2O2.
- Move the image files from both experimental dates into the appropriate folder according to the experimental condition in the file name.
- Ensure that all of your images from one experimental condition are in one folder together.
- Download AnalyzeH2AX_FITCintensityBatch_Fa20script (linked here).
- Right click on the link and download the file into a folder where you can find it.
- In ImageJ, go to Plugins--> Macros--> Run, and click on the AnalyzeH2AX_FITCintensityBatch_Fa18 script that you downloaded.
- When the script prompts you to "Choose input folder," choose the folder containing all your .tif image stacks (folder named by one experimental condition), and click "Open."
- In the dialog box titled "Choose Intensity Threshold Values," type in the corresponding DAPI threshold values you have chosen, and click "OK."
- You will be prompted to name the resulting Excel file next.
- Please wait for the script to run through all your images for one condition. In the end all the image files will pop up, along with the "drawings" that show where it identified cells in your images.
- The script will output one Excel file into your image folder.
- Before closing any images, validate the results in the Excel file with the images in ImageJ.
- Choose a few representative images to verify.
- Check the "Drawing" images and DAPI images to see if the nuclei were called correctly by your threshold values.
- Repeat this analysis for each folder containing images from an experimental condition.
- To calculate total intensity of FITC signal per nucleus divide the RawIntDens (sum of the intensity of all the pixels in arbitrary units, a.u.) by area (pixels2) for each outline that represents a nucleus.
- Finally, calculate the average of the total FITC intensity(a.u.) per area (px2) for all nuclei in each experimental condition.
Part 5: Analyze γH2AX images by counting foci
In addition to the above analysis, you will also use ImageJ to enumerate the γH2AX foci present in the nuclei of the treated cells. To do this use the protocol written by researchers at Duke University. Follow the direction outlined here to analyze your images.
In your laboratory notebook, complete the following:
- How does the data obtained in the two analysis approaches compare? Are the results the same? Different?
- Which analysis approach best represents the raw data images? Why?
Next day: Treat cells and perform high-throughput genome damage assay
Previous day: Use immunoflourescence staining to assess repair foci experiment