|
|
| (65 intermediate revisions by 3 users not shown) |
| Line 3: |
Line 3: |
| | [[Category:Optical Microscopy Lab]] | | [[Category:Optical Microscopy Lab]] |
| | {{Template:20.309}} | | {{Template:20.309}} |
| − | ===Overview===
| + | __NOTOC__ |
| | | | |
| − | <blockquote>
| |
| − | <div>
| |
| − | ''I took a good clear piece of Cork, and with a Pen-knife sharpen'd as keen as a Razor, I cut a piece of it off, and thereby left the surface of it exceeding smooth, then examining it very diligently with a Microscope, me thought I could perceive it to appear a little porous; but I could not so plainly distinguish them, as to be sure that they were pores, much less what Figure they were of: But judging from the lightness and yielding quality of the Cork, that certainly the texture could not be so curious, but that possibly, if I could use some further diligence, I might find it to be discernable with a Microscope, I with the same sharp Penknife, cut off from the former smooth surface an exceeding thin piece of it, and placing it on a black object Plate, because it was it self a white body, and casting the light on it with a deep plano-convex Glass, I could exceeding plainly perceive it to be all perforated and porous, much like a Honey-comb, but that the pores of it were not regular; yet it was not unlike a Honey-comb in these particulars.''
| |
| | | | |
| − | ''I told several lines of these pores, and found that there were usually about threescore of these small Cells placed end-ways in the eighteenth part of an Inch in length, whence I concluded there must be neer eleven hundred of them, or somewhat more then a thousand in the length of an Inch, and therefore in a square Inch above a Million, or 1166400. and in a Cubick Inch, above twelve hundred Millions, or 1259712000. a thing almost incredible, did not our Microscope assure us of it by ocular demonstration.''
| + | ==Measure the microscope's magnification == |
| | | | |
| − | <blockquote> | + | <figure id="fig:Manta_camera_side_view"> |
| − | ''— [http://en.wikipedia.org/wiki/Robert_Hooke Robert Hooke] from Micrographia: or Some Physiological Descriptions of Minute Bodies made by Magnifying Glasses with Observations and Inquiries Thereupon (1665)<ref name="Micrographia">Hooke, R. [http://www.gutenberg.org/files/15491/15491-h/15491-h.htm Micrographia: or Some Physiological Descriptions of Minute Bodies made by Magnifying Glasses with Observations and Inquiries Thereupon] London:Jo. Martyn, and Ja. Allestry, Printers to the Royal Society; 1665</ref> ''
| + | [[Image:20.309_130813_BrightFieldExampleImages.png|right|thumb|Example images included by past students in their Week 1 report: (top) Air Force target, (center) Silica spheres and dust, (bottom) Ronchi Ruling]]</figure> |
| − | </blockquote>
| + | |
| − | </div>
| + | |
| − | </blockquote>
| + | |
| − | <br/> | + | |
| | | | |
| − | ====Microscope block diagram====
| + | Measuring the magnification of your microscope is a good way to verify that your instrument has been constructed correctly. (In general, you should measure the magnification of any microscope you will use to make quantitative measurements of size). Use the measured value in your calculations, not the number printed on the objective. |
| − | To get you started, here is a block diagram of a 20.309 microscope. Note that for Assignment 1, you will be building the brightfieqld (or transillumination) path. You should leave space for the fluorescence path, which you will complete next week.
| + | |
| | | | |
| − | [[Image:20.309 130911 YourMicroscope.png|center|thumb|400px|20.309 microscope block diagram]] | + | # Install the 10x objective in your microscope and image the microruler calibration slide. |
| | + | #* You will find the objectives in the west drawers of the lab. Be sure to pick up a RMS to SM1 adaptor from the neighboring bin to be able to thread the objective into the Thorlabs cage cube. |
| | + | #* The microruler calibration slide has tick marks that are 10 μm apart. Every 100 μm, there is a longer tick mark. |
| | + | #* Make sure that the side of the microruler with the pattern on it faces the objective. Imaging through the thick glass causes distortion and many other troubles. |
| | + | # Start the live preview using the <tt>UsefulImageAcquisition</tt> tool: |
| | + | #* Familiarize yourself with the <tt>UsefulImageAcquisition</tt> tool and how to save and display images by reading through [[Recording, displaying and saving images in MATLAB|these instructions]]. |
| | + | # Ensure that the camera's field of view is approximately centered in the objective's field of view. |
| | + | #* The objective has a larger field of view (FOV) than the camera. Adjust the 45 degree mirror to traverse the objective's FOV horizontally and vertically. The FOV is approximately circular. Find a spot near the middle. |
| | + | # Record an image of the microruler. |
| | + | #* Once your happy with the image on the live preview, click the <tt>Acquire</tt> button. |
| | + | #* Display the image and use <tt>imdistline</tt> or the data cursor to measure a known distance between rulings in your image and compute the magnification. |
| | + | #* When choosing a distance to measure, consider the factors that influence the uncertainty of your measurement. |
| | + | # '''Save your images in a .mat file''' for later use in MATLAB or as a PNG image for use in your report or other programs (Did you read [[Recording, displaying and saving images in MATLAB| the instructions??]]). |
| | + | # Repeat the magnification measurement for the 40x and 100x objectives. |
| | + | # Using your magnification measurements and the known size of the CMOS camera, calculate the field of view (FOV) of the microscope for each objective. |
| | | | |
| − | ====Optical components==== | + | {{Template:Assignment Turn In|message=<br /> |
| − | Below is a brief introduction to a few of the different components comprising your microscope. Various systems for optical construction are available based on rails, posts, cages, tubes, and all manner of little, metallic bits. The 20.309 microscope is constructed chiefly from cage and lens tube components made by a company called [http://www.thorlabs.com/ ThorLabs]. The structure should be rigid, and the components sufficiently tightened so that your optics remain aligned even after moving your microscope. Understanding how all of the components work together can be daunting. Ask about any components that perplex you.
| + | * Display an example image of the ruler at each magnification, and |
| | + | * Make a table displaying the manufacturer specified magnification (i.e. the printed number on the objective), the as-designed magnification (based on the 125 mm tube lens), the object height, the image height, the measured magnification and the FOV (see example below). Don't forget to include appropriate units. Report the length and width of the FOV of the camera (in distance units), not its area (in distance units squared). |
| | + | * Describe how you chose the distance to measure in your images. |
| | + | }} |
| | + | {| class="wikitable" style="text-align: center" |
| | + | |- |
| | + | | Manufacturer specified magnification |
| | + | | As-designed magnification |
| | + | | Object height (μm) |
| | + | | Image height (μm) |
| | + | | Measured magnification |
| | + | | Field of view (FOV, μm x μm) |
| | + | |- |
| | + | | 10x || || || || || |
| | + | |- |
| | + | | 40x || || || || || |
| | + | |- |
| | + | | 100x || || || || || |
| | + | |} |
| | | | |
| − | =====Lenses===== | + | ==Measure particle size== |
| | | | |
| − | Plano-convex spherical lenses are available with focal lengths of 25, 50, 75, 100, 125, 150, 175, and 200 mm. Plano-concave lenses with focal lengths of -30 and -50 are also available. It is best to mount most optics in short (''e.g.'' 0.5") lens tubes. It is acceptable to mount a lens between the end of a tube and a tube ring or between two tube rings. In most cases, the convex side of the lens faces toward the collimated beam; the planar side goes toward the convergent rays.
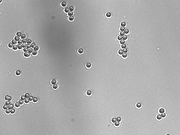
| + | [[Image:20.309_130813_BF_3p2umbeads_40x.png|right|thumb|Example image of 3.2 μm beads using the instructor microscope. Submit picture to replace this!]] |
| | | | |
| − | * ''Tip:'' Verify all optics before you use them by determining the focal length with a ruler. You can use the lens measuring station. Alternatively, you can use the ceiling fluorescent lamps as a light source and measure the exact distance between the lens(es) assessed and the lamp's image. Can you imagine a simple rig to evaluate negative focal lengths (of plano-concave lenses for instance)?
| + | Now that you know the magnification of your instrument, use it to measure the size of some microscopic objects as imaged with the 40x objective lens only. Slides with 7.2 μm and 3.2 μm silica microspheres are available in the lab. |
| − | * ''Tip:'' As you install lenses into your microscope, put a piece of tape on the lens tube showing focal length and orientation. This will help you both during construction and put-away. Save the lens storage boxes and return components to the correct boxes when you are done.
| + | |
| − | * Handle lenses only by the edges. If a lens is dirty, first remove grit with a blast of clean air or CO<sub>2</sub>. Clean the lens by wiping with a folded piece of lens paper wetted with a drop of methanol. (Do not touch the part of the tissue you use for cleaning with your fingers.) In some cases, it may be helpful to hold the folded lens tissue in a hemostat. Ask an instructor if you need help.
| + | |
| | | | |
| − | =====Objective lenses=====
| + | * Image 7.2 μm and 3.2 μm silica microspheres using the 40x objective only. |
| | + | * Measure the size of several beads of each type. |
| | + | * Try to measure beads that are relatively sparse or not too clumped together. |
| | | | |
| − | Please see the Nikon [http://www.microscopyu.com/articles/optics/objectiveintro.html Introduction to Microscope Objectives] at their excellent [http://www.microscopyu.com/index.html MicroscopyU] website.
| + | How many spheres should you measure? |
| | + | * One typical way to estimate the uncertainty in a measurement is to make many measurements of the same thing, and then calculate the standard error. Standard error is equal to the sample standard deviation divided by the square root of the number of data points. It is an estimate of how well you know the mean value of whatever you are measuring. Assuming nothing is changing with your measurement over time, increasing the number of data points will reduce your standard error. On the flip side, you cannot sit in lab forever and make an infinite number of measurements to reduce your uncertainty to zero. What do you think should determine how many measurements you take? |
| | | | |
| − | There are three objective lenses available in the lab: a 10×, a 40×, and a 100×. All of these are designed to use a 200 mm tube lens to form an image on the camera. An adapter ring converts the objective mounting threads to the SM1 threads used by the lens tube system.
| + | Reporting uncertainty: |
| | + | * All measured values should be reported with an associated measure of variability, which is usually the range, standard deviation, or standard error of the dataset. Use the abbreviation "s.d." for standard deviation and "s.e.m." for standard error after the "±". For example: 1.21 ± 0.03 GW (±s.d., ''N''=42). Uncertainty is typically reported with one or two significant figures. Round uncertain quantities to the same decimal place as the uncertainty. The sample size must be included in all cases. Report uncertainty in the same units as the measurand. <ref>Relative (percent) uncertainty is undesirable in the presence of additive noise because a constant magnitude error source produces different error values as the measurand changes. Relative error is a more sensible choice when measuring an unvarying quantity such as a physical constant.</ref> Standard error is typically the best choice for datasets that contain 20 or more samples. It can be interpreted as an estimate of the size of an interval that would contain the result of repeating the experiment about ⅔ of the time. Range or standard deviation is a good choice for small values of ''N''. |
| | | | |
| − | [[Image:20.309_130813_SimpleMicroscopeDiagram.png|center|thumb|400px|The reference tube length for the Nikon objectives we will use is 200 mm. A 200 mm lens, placed 200 mm from the CCD, will produce the rated magnification M.]]
| + | {{Template:Assignment Turn In|message=<br /> |
| − | * ''Working distance'' (WD) is the distance between the front objective lens surface and the cover slip, and so it is also approximately the distance to the front focal plane. In order to focus an image at the back focal plane of the tube lens, i.e., on the CCD array, the sample plane must coincide with the front focal plane in a 4f microscope arrangement. The stage is added to hold the sample in this plane.
| + | # Display an example image of each bead size. |
| − | * The 100× objective is designed to be used with immersion oil. When using the 100× objective, place a drop of oil directly on the tip on the objective. Bring the drop in contact with the slide cover glass. After use, clean off the remaining oil by wicking it away with lens paper or a Kim-wipe. Do not put samples away dirty.
| + | # Report the average size and uncertainty of the spheres in each sample, (be sure to include the number of samples measured). |
| − | * Note that the back focal plane (BFP) of the objective does not necessarily coincide with the rear of the objective housing. In fact, for the Nikon 40x objective the BFP is close to the blue ring. You will find its actual location when aligning the laser path in Part 2. The 200 mm distance labeled between the back of the objective housing and the tube lens is a recommendation from Nikon to enable optimal imaging. For details on the importance and origin of this distance please ask an instructor.
| + | # Discuss how the measured bead sizes compared to the nominal size. |
| − | | + | # In one or two sentences, explain how you chose the number of samples to measure. }} |
| − | =====Sample stage=====
| + | |
| − | | + | |
| − | A precision Newport X/Y/Z stage<ref>[http://www.newport.com/562-Series-ULTRAlign-Precision-Multi-Axis-Positio/140089/1033/catalog.aspx Precision Newport X/Y/Z stages]</ref> with a sample holder mounted on a post, or a Thorlabs Max312D stage, also with a sample holder, is available at each lab station. The Newport stage setup is top-heavy. Avoid accidents by ensuring that the post base is always attached to an optical breadboard or table. Leave the stage at the lab station when you are done with it. For the Thorlabs stages, it is still a good idea to bolt them down so that your area of interest (AOI) stays in your microscope field of view (FOV).
| + | |
| − | | + | |
| − | All stage axes have limited adjustment range, especially the Thorlabs stages. To deal with this, it is best to leave the stage base bolts and sample holder bolts loose and move the sample holder in x, y and z to roughly find your AOI. Once you are on or near your AOI, tighten the bolts and use the micrometers to center your image. One trick here is to get the z clamped first, then deal with x and y.
| + | |
| − | | + | |
| − | =====CCD camera=====
| + | |
| − | | + | |
| − | The microscope you will build does not have an eyepiece for direct visual observation. Instead, images will be captured with a CCD camera<ref>[http://www.alliedvisiontec.com/emea/products/cameras/gigabit-ethernet/manta/g-032bc.html Allied Manta G032B]</ref>. Its monochrome (black and white) sensor contains a grid of 656×492 square pixels that measure 7.4 μm on a side. An adapter ring converts the C-mount thread on the camera to SM1.
| + | |
| − | | + | |
| − | ====Design====
| + | |
| − | | + | |
| − | Sketch out the design for your microscope on paper (or print out the above diagram). For this part of the lab, you can just draw the bright field illumination path. Label all the optical elements (lenses, mirrors, microscope objectives, and camera), distances, lens specifications, and orientations.
| + | |
| − | * Some elements must be positioned precise distances apart; other distances are not critical. Use ray-tracing to determine when this is the case. Which distances in your bright-field microscope will be critical? Which will be forgiving or unessential? Which will change with each objective lens (10×, 40× and 100×)?
| + | |
| − | * Which sections of the light path can be open (strut-based structure, cage rods)? Which would better enclosed (Thorlabs lens tubes)?
| + | |
| − | * In what way will the illumination LED color affect your design? your results?
| + | |
| − | * Which lens will you use between the LED and the sample for bright field transmitted light imaging?
| + | |
| − | | + | |
| − | Next, take a look at the example microscope in the lab. This microscope is here for your reference, but please do not touch, alter, or remove parts from the example microscope.
| + | |
| − | [[Image:130814_Microscope_All.jpg|center|thumb|400px|The general layout of the 20.309 microscope is compact and stand-alone; it fits and can be transported onto a breadboard]]
| + | |
| − | | + | |
| − | Can you identify all the components of your diagram? Try to think about the purpose of each component, and why it is laid out a particular way. Once you're satisfied that you understand the big picture, it's time to build your own!
| + | |
| − | | + | |
| − | A few tips to keep in mind:
| + | |
| − | * Reproduce the general layout of the example microscope: it grants compactness and allows your device to be a stand-alone breadboard-transportable microscope.
| + | |
| − | * Even though you're first focusing on the bright-field imaging leg of your microscope, take into consideration some requirements pertinent to the fluorescence imaging elements you'll add to your system next week:
| + | |
| − | | + | |
| − | ===Exercise 1: Assemble the microscope===
| + | |
| − | | + | |
| − | ====Assemble the base====
| + | |
| − | Gather the following parts:
| + | |
| − | <gallery widths=216px caption="Optical breadboard(Located in the lower left cubby of the Instructor Cubbies):">
| + | |
| − | File:OpticalBreadBoard.jpg|Optical breadboard
| + | |
| − | File:VerticalMountingPost.jpg|Vertical Thorlabs P14 mounting post (1.5" diameter)
| + | |
| − | </gallery>
| + | |
| − | | + | |
| − | * On a 1' x 2' x <sup>1</sup>/<sub>2</sub>" optical breadboard, align the vertical Thorlabs P14 (1.5" diameter mounting post) with a breadboard hole that is 11 positions from a short side and 5 positions from a long side. This allows enough free space on the breadboard such that either the Newport or the Thorlabs stages may be utilized. (Note that the above picture is in error as the P14 is only 9 positions from a short side of the breadboard.)
| + | |
| − | | + | |
| − | ====Assemble the illuminator====
| + | |
| − | Gather parts:
| + | |
| − | <gallery widths=216px caption="Optomechanics and LED (located in plastic bins on top of the center parts cabinet):">
| + | |
| − | File:LensTube05.jpg|1 x 0.5" Lens tube (SM1L05)
| + | |
| − | File:LCP01.jpg|1 x 2" Cage plate (LCP01, looks like an "O" in a square)
| + | |
| − | File:LCP02.jpg|1 x Cage plate adapter (LCP02, looks like an "X")
| + | |
| − | File:SM2RR.jpg|2 x 2" Retaining rings (SM2RR)
| + | |
| − | File:RedLED.jpg|1 x red, super-bright LED (mounted in heatsink)
| + | |
| − | </gallery>
| + | |
| − | | + | |
| − | <gallery widths=216px caption="Optomechanics (located on the counter above the west drawers):">
| + | |
| − | File:ER8.jpg|3 x ER2 cage assembly rod (The last digit of the part number is the length in inches.)
| + | |
| − | File:SM1RR.jpg|1 x 1" Retaining rings (SM1RR)
| + | |
| − | </gallery>
| + | |
| − | | + | |
| − | <gallery widths=216px caption="Optics (located in the west drawers):">
| + | |
| − | File:Lenses.jpg|1 x LA1951 plano-convex, f = 25 mm lens (this will be used as a condenser for your illuminator)
| + | |
| − | </gallery>
| + | |
| − | | + | |
| − | Most of the tools you will need are located in the drawers next to your lab station. Hex keys (also called Allen wrenches) are used to operate SHCSs. Some hex keys have a flat end and others have a ball on the end, called balldrivers. The ball makes it possible to use the driver at an angle to the screw axis, which is very useful in tight spaces. You can get things tighter (and tight things looser) with a flat driver. Here is a list of the tools you will need:
| + | |
| − | | + | |
| − | <gallery widths=216px caption="Tools (located in your station's drawers):">
| + | |
| − | File:BallDrivers.jpg|1 x 3/16 hex balldriver for 1/4-20 cap screws <br/>1 x 9/64 hex balldriver, <br/>1 x 0.050" hex balldriver for 4-40 set screws (tiny)
| + | |
| − | File:SPW602.jpg|1 x SPW602 spanner wrench
| + | |
| − | </gallery>
| + | |
| − | | + | |
| − | You will also need to use an adjustable spanner wrench. The adjustable spanner resides at the lens cleaning station. There are only one or two of these in the lab. It is likely that one of your classmates neglected to return it to the proper place. This situation can frequently be remedied by yelling, "who has the adjustable spanner wrench?" at the top of your lungs. Try not to use any expletives. And please return the adjustable spanner wrench to the lens cleaning station when you are done.
| + | |
| − | | + | |
| − | <gallery widths=216px>
| + | |
| − | File:SPW801.jpg|1 x SPW801 adjustable spanner wrench
| + | |
| − | </gallery>
| + | |
| − | | + | |
| − | * Mount the LED between two SM2RR retaining rings in an LCP01 cage plate.
| + | |
| − | ** Screw in one SM2RR to a depth of 1 mm.
| + | |
| − | ** Run the wires of the LED through the opening in the LCP01 and insert the LED until it is resting on the retaining ring. It will get sandwiched in-between two retaining rings.
| + | |
| − | ** Add a second SM2RR retaining ring to secure the LED. Use the SPW801 adjustable spanner wrench or a small flat bladed screwdriver to tighten the retaining ring. The SPW801 can be opened until its width matches the SM2RR diameter, the separation between the ring's notches.
| + | |
| − | <center>
| + | |
| − | [[Image: 140729_OpticsBootcamp_05.jpg|frameless|x200px]]
| + | |
| − | [[Image: 140729_OpticsBootcamp_07.jpg|frameless|x200px]]
| + | |
| − | </center>
| + | |
| − | | + | |
| − | * Thread an SM1RR retaining ring in another SM1L05 lens tube and use the SPW602 spanner wrench to drive it about 90% of the way down the tube.
| + | |
| − | * Place the 25 mm lens in the SM1L05 lens tube with its curved side facing the external threads of the tube.
| + | |
| − | ** Don't just drop the lens in. Use lens paper to gently lower the lens into the tube.
| + | |
| − | ** Don't touch the lens while you are putting it in.
| + | |
| − | * Thread a second SM1RR retaining ring into the lens tube and tighten it with the SPW602 spanner wrench.
| + | |
| − | <center>
| + | |
| − | [[Image: LensInLensTube.JPG|frameless|x200px]]
| + | |
| − | </center>
| + | |
| − | | + | |
| − | *Screw the lens tube with the 25 mm lens a into an LCP02
| + | |
| − | <center>
| + | |
| − | [[Image: LensTubeLCP02.JPG|frameless|x200px]]
| + | |
| − | </center>
| + | |
| − | | + | |
| − | * Connect the LCP02 and the LCP02 together using cage rods
| + | |
| − | | + | |
| − | *Connect the LED
| + | |
| − | ** A red or a blue LED illuminator can be used for bright-field transmitted light imaging. On one hand a blue LED yields a better bright-field resolution, however bright-field resolution is not usually critical in this lab. On the other hand, a red LED allows simultaneous fluorescent and bright-field imaging in Parts 2 and 3, and this can be quite useful when trying to bring a fluorescent sample into focus.
| + | |
| − | ** Turn the power supply on.
| + | |
| − | ** Make sure the power supply is not enabled (green LED below the OUTPUT button is not lit).
| + | |
| − | ** Use the righthand set of knobs to set the current and voltage
| + | |
| − | *** Adjust the CH1/MASTER VOLTAGE knob so the display reads about 5 Volts.
| + | |
| − | *** Adjust the CH1/MASTER CURRENT knob to so the display reads 0.1 Amps.
| + | |
| − | *** IMPORTANT: Never set the CURRENT to a value greater than 0.5A, as this will burn out the LED.
| + | |
| − | ** Connect the + (red) terminal of channel CH1 on the power supply to the red wire of the LED.
| + | |
| − | ** Connect the - (black) terminal of channel CH1 on the power supply to the black wire of the LED.
| + | |
| − | ** Press the OUTPUT button to enable the power supply and light the LED.
| + | |
| − | ** Adjust the LED brightness using the power supply's CURRENT knob.
| + | |
| − | <center>
| + | |
| − | [[Image: 140730_OpticsBootcamp_1.jpg|frameless|x200px]]
| + | |
| − | [[Image: 140730_OpticsBootcamp_2.jpg|frameless|x200px]]
| + | |
| − | </center>
| + | |
| − | | + | |
| − | {{Template:Safety Warning|message=Double check your wiring before powering the LED. The LED can be damaged by excessive current. Limit the driving current to 0.5 A to protect the LED.}} | + | |
| − | | + | |
| − | *Collimate the illuminator
| + | |
| − | **Slide the lens along the cage rods until the light from the LED focuses at some point far away (like on the wall)
| + | |
| − | **Tighten the set screws to keep the lens a fixed distance from the LED. There is no need to overnighted the screws, just make sure that the lens doesn't slide around or move relative to the LED.
| + | |
| − | | + | |
| − | ====Assemble the objective cage components ====
| + | |
| − | *** Set the distance between the top of the breadboard and the top surface of the upper LCP01 to '''13.5 cm'''. It is important to ensure your construction is compatible with either of the two distinct stage mounting platforms available in the 20.309 lab (either Newport or Thorlabs model). If you find it inconvenient to measure this, there is a Handy Scope Height Thingama-jig floating around the lab. Ask your instructor(s). Also, note that the stages are very expensive; always lift them from the bottom.
| + | |
| − | ***
| + | |
| − | | + | |
| − | ** Do insert the C6W cage cube that will later hold the dichroic mirror on while fluorescence imaging will rely. Be sure to keep the mounting struts fully recessed in the cube walls; their ends should not stick out, they would otherwise hinder maneuvers with dichroic-holding kinematic plate!
| + | |
| − | [[Image:130816_CageCube.png|center|thumb|400px|The mounting struts should remain recessed within the cage cube walls.]]
| + | |
| − | | + | |
| − | * Verify the focal length of the lenses you selected. If you find an optic in the wrong box: identify the optic and replace it in the correct box or label the box correctly. (Ask an instructor if you can't find the right box. There are many boxes near the wire spools behind you as you stand at the wet bench.)
| + | |
| − | * Check all your lenses for cleanliness before you use them. You'll save yourself some troubleshooting time and effort down the road!
| + | |
| − | * Make sure all your components are "leveled" (horizontal, not slanted).
| + | |
| − | * Use tube rings (and never an SM1T2, SM1V01, or SM1V05) to mount optics in lens tubes.
| + | |
| − | * Use adjustable mounting components in front of the CCD camera so you can optimize and fine-tune the camera positing with respect to the imaging lens ''L2''. Beware: never use an SM1T2 coupler without a locking ring — they are very difficult to remove if they are tightened against a lens tube or tube ring. Also put a quick-connect in your design such that the camera CCD will end up 200 mm from the back focal plane of the objective. Remember that the CCD is recessed inside the opening of the camera.
| + | |
| − | [[Image:20.309 130816 CCD QuickConnect.png|center|thumb|400px|Adjustable Thorlabs SM1V05 and SM1T2 connectors precede the quick-connect union to the CCD camera.]]
| + | |
| − | * Use only three cage rails to connect the C6W cage cube and the KCB containing the last silver mirror before the CCD camera, so you can easily take in and out the barrier filter (BF) that will later aid fluorescence-mode microscopy. Always place two rails at the top so that an alignment target can be hung if needed (the benefit will become more clear in Part 2).
| + | |
| − | | + | |
| − | [[Image:20.309 130816 BarrierFilterSpace.png|center|thumb|250px| Insertion and removal of optical components is facilitated by a three-strut-only link. ]]
| + | |
| − | | + | |
| − | * The Nikon objective lenses are designed to be paired with a 200 mm tube lens.
| + | |
| − | * Assume that the objectives behave as ideal plano-convex lenses.
| + | |
| − | * Fine focusing will be achieved by adjusting the height of the sample stage.
| + | |
| − | * ''Tip:'' Throughout the optical microscopy lab, start the alignment with a 10× objective and then progress to 40× and 100×.
| + | |
| − | | + | |
| − | | + | |
| − | ====Assemble the remaining beam path====
| + | |
| − | ====Putting it all together====
| + | |
| − | | + | |
| − | <div id="MATLABsaving"></div>
| + | |
| − | ====Recording, displaying and saving images in MATLAB====
| + | |
| − | <figure id="fig:UsefulImageAcquisition">
| + | |
| − | [[Image:UsefulImageAcquisiton.png|thumb|right|<caption>The UsefulImageAquisition window can be used to (hopefully) easily control the camera settings. To run it, type "<tt>foo = UsefulImageAcquisition;
| + | |
| − | foo.Initialize</tt>" into the MATLAB console window.</caption>]]
| + | |
| − | </figure>
| + | |
| − | | + | |
| − | <ol>
| + | |
| − | <li> Run the UsefulAcquisition tool
| + | |
| − | <ul> <li> Launch MATLAB and in the command window type: </li>
| + | |
| − | <pre>foo = UsefulImageAcquisition;
| + | |
| − | foo.Initialize </pre>
| + | |
| − | <li> The Image Acquisition window (<xr id="fig:UsefulImageAcquisition" />) will open with the controls for the camera.</li>
| + | |
| − | <li> The "Manta_G-032B" camera is configured to produce 12-bit, monochrome images. In this mode, the intensity of each pixel in the image will be represented by 12 binary digits, allowing a range of values from 0-4095.</li>
| + | |
| − | <li> In the Image Acquisition window set "Frame Rate" to 20. This will cause the camera to take 20 complete images per second (this will only be relevant when recording movies in Part 3 of the lab).</li>
| + | |
| − | <li> Click the "Start Preview" button. The live image from the camera should appear in the Preview pane.</li>
| + | |
| − | <li> If this does not produce a live image, use an appropriate expletive, and ask an instructor to figure out what the heck went wrong.</li>
| + | |
| − | <li> Change the "Exposure" setting to produce a good image. The value sets the exposure time for each frame in microseconds. </li>
| + | |
| − | </ul></li>
| + | |
| − | <li> Recording an image
| + | |
| − | <ul><li> In the Image Acquisition window, set "Number of Frames" to 1. This setting controls how many images MATLAB will record each time you press the "Acquire" button.</li>
| + | |
| − | <li> When you are happy with the image displayed by the live preview, press "Acquire". The live preview will stop.</li>
| + | |
| − | <li> The image is now stored in the <tt>foo.ImageData</tt> variable, which will update each time you acquire a new image. To copy the data into a new variable, choose a descriptive name for your image like 'microruler10x' and save it to your workspace by typing the following into the MATLAB command window:</li>
| + | |
| − | <pre>microruler10x = foo.ImageData;</pre>
| + | |
| − | <li> Next, in the MATLAB command window type </li>
| + | |
| − | <pre>whos microruler10x </pre>
| + | |
| − | <li> This command will display relevant information about the new variable you’ve created. You should see that the image is represented as a 492x656 matrix of 16-bit integers.</li>
| + | |
| − | </ul></li>
| + | |
| − | <li> Displaying the image
| + | |
| − | <ul><li> You can display images using a variety of commands in MATLAB. In the optics bootcamp we used the <tt>imagesc</tt> command which scales the image intensity (the brightest pixel is white, the darkest is black). In some cases, like when you have very dim images, this command can be misleading. It’s better to use the un-scaled <tt>imshow</tt> command for quantitative measurements. </li>
| + | |
| − | <li> When the 12-bit numbers from the camera get transferred to the computer, they are converted to 16-bit numbers. 16-bit numbers can represent a range of values from 0-65535, while your 12-bit image only contains values from 0-4095. This leaves a considerable portion of the number range unoccupied. Consequently, if you type <tt>imshow( microruler10x )</tt>, you will see an image that looks almost completely black (try it!). </li>
| + | |
| − | <li> One way to make this work is to rescale your image to 16 bits: <tt> imshow( 65535/4095 * microruler10x )</tt></li>
| + | |
| − | <li> An even better way to work with images in MATLAB is to convert them to [http://en.wikipedia.org/wiki/Double-precision_floating-point_format double precision floating point format] straightaway. Double precision floating point numbers can represent an extremely wide range of values with high precision. Convert the image to a double and rescale it using the following command:</li>
| + | |
| − | <pre>microruler10x = double( microruler10x ) / 4095; </pre>
| + | |
| − | <li> This conversion has made your image into a double with a range of intensities from 0-1, with 1 being full intensity and 0 completely dark. Now try: </li>
| + | |
| − | <pre>whos microruler10x
| + | |
| − | imshow( microruler10x ) </pre>
| + | |
| − | </ul></li>
| + | |
| − | <li> Saving your image
| + | |
| − | <ul><li>Save images in a .mat format so that you can easily reload them into Matlab for later use. </li>
| + | |
| − | <pre>
| + | |
| − | save microrulerImages % saves entire workspace to filename 'microrulerImages.mat'
| + | |
| − | save microrulerImages image1 image2 %saves only variables image1 and image2 to filename 'microrulerImages.mat'</pre>
| + | |
| − | <li> To reload your data the next time you open matlab, navigate to the folder where you saved your workspace, type </li>
| + | |
| − | <pre>load microrulerImages </pre>
| + | |
| − | <li> If you want to save individual images as a .PNG (a good format for use in your report or other programs), the command might look something like: </li>
| + | |
| − | <pre>imwrite( im2uint16( microruler10x ), 'microruler10x.png', 'png' );</pre>
| + | |
| − | <li> Note that you can also use the File→Save As menu after displaying an image or figure. This is useful if you want to save additions to your images (like data cursors). However, we recommend always saving the raw data as .mat files so that you are able to re-process your images later.
| + | |
| − | </ul></li>
| + | |
| − | </ol>
| + | |
| − | | + | |
| − | ===Exercise 2: Measure the microscope's magnification ===
| + | |
| − | <figure id="fig:Manta_camera_side_view">
| + | |
| − | [[Image:20.309_130813_BrightFieldExampleImages.png|right|thumb|Example images included by past students in their Week 1 report: (top) Air Force target, (center) Silica spheres and dust, (bottom) Ronchi Ruling]]
| + | |
| − | | + | |
| − | Measuring the magnification of your microscope is a good way to verify that your instrument is functioning well. You should measure the magnification of any microscope you plan to use for making quantitative measurements of size. Use the measured value in your calculations, not the number printed on the objective. Consider the uncertainty in your measurement.
| + | |
| − | # Start the live preview using the UsefulImageAcquisition tool | + | |
| − | # Ensure that the camera's field of view is approximately centered in the objective's field of view.
| + | |
| − | #* The objective has a larger FOV than the camera. Use the adjustment knobs on mirror M1 to traverse the objective's FOV horizontally and vertically. The FOV is approximately circular. Find a spot near the middle.
| + | |
| − | # Start with the 10x objective and a microruler calibration slide.
| + | |
| − | #* The microruler calibration slide has tick marks that are 10 um apart. Every 100 um, there is a longer tick mark.
| + | |
| − | #* Make sure that the side of the microruler with the pattern on it faces the objective. Imaging through the thick glass causes distortion and many other troubles.
| + | |
| − | # Record an image of the microruler.
| + | |
| − | # Use <tt>imdistline</tt> or the data cursor to measure a known distance between rulings in your image and compute the magnification.
| + | |
| − | #* When choosing a distance to measure, consider the factors that influence the uncertainty of your measurement.
| + | |
| − | # Repeat the magnification measurement for the 40x and 100x objectives.
| + | |
| − | #* With the 100x objective, you may want to substitute the microruler with a Ronchi Ruling, a grating with 600 line pairs per millimeter. Why is it not wise to use the Ronchi Ruling with the 10x objective?
| + | |
| − | # Save your images in a .mat file for later use in MATLAB or as a PNG image for use in your report or other programs.
| + | |
| − | # Using your magnification measurements, calculate the FOV of the microscope for each objective.
| + | |
| − | | + | |
| − | ===Exercise 3: Particle size measurement===
| + | |
| − | [[Image:20.309_130813_BF_3p2umbeads_40x.png|right|thumb|Example image of 3.2 μm beads using the instructor microscope. Submit picture to replace this!]]
| + | |
| − | | + | |
| − | Now that you know the magnification of your instrument, use it to measure the size of some microscopic objects as imaged with the 40x objective lens only. Slides with 7.2 μm, 3.2 μm and 1 μm silica microspheres are available in the lab.
| + | |
| − | | + | |
| − | # Image 7.2 μm, 3.2 μm and 1 μm silica microspheres as described in the magnification measurement procedure (40x objective only).
| + | |
| − | # Measure and report the average size and uncertainty of the spheres in each sample. How many spheres should you measure?
| + | |
| | | | |
| | ==Microscope storage== | | ==Microscope storage== |
| Line 273: |
Line 78: |
| | * Keep all of the boxes for the optics you use with your instrument to simplify putting things away. | | * Keep all of the boxes for the optics you use with your instrument to simplify putting things away. |
| | * Take a blue bin to store loose items (such as lens boxes) in. | | * Take a blue bin to store loose items (such as lens boxes) in. |
| − | * '''Stages, CCD cameras, neutral density filters and barrier filters stay at the lab station'''. Do not store these with your microscope. | + | * '''Stages, CCD cameras, neutral density filters and emission filters stay at the lab station'''. Do not store these with your microscope. |
| | * Return objective lenses to the drawer when you are not using them. (Do not store them with your microscope.) | | * Return objective lenses to the drawer when you are not using them. (Do not store them with your microscope.) |
| | * The stages are very expensive. Always lift from the bottom. | | * The stages are very expensive. Always lift from the bottom. |
| | * If you break something (or discover something pre-broken for you), do not return it to the component stock. Give all broken items to an instructor. You will not be penalized for breaking something, but not reporting may be looked upon less kindly. | | * If you break something (or discover something pre-broken for you), do not return it to the component stock. Give all broken items to an instructor. You will not be penalized for breaking something, but not reporting may be looked upon less kindly. |
| | + | |
| | + | ==References== |
| | + | <references /> |
| | + | |
| | + | {{Template:Assignment 1 navigation}} |
| | | | |
| | {{Template:20.309 bottom}} | | {{Template:20.309 bottom}} |