20.109(S21):M3D5
Contents
Introduction
Protocols
- As you consider sites that may alter calcium binding, keep the following in mind:
- When this module was first debuted, everyone mutated residues directly in the calcium binding loops, and very few groups saw dramatic changes in affinity or cooperativity of calmodulin with respect to calcium.
- In some years, class-wide results suggested that mutations in the first two binding loops were more likely to have an effect than mutations in the latter two binding loops.
- Some folks also targeted non-binding structural areas, but results were inconclusive.
- You may repeat or otherwise build upon prior results as long as you give your own reasoning.
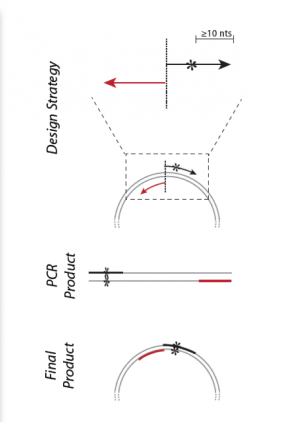
Part 3: Primer design for mutagenesis
It wouldn’t be very experimentally efficient to somehow pick out and modify a single residue on inverse pericam post-translationally. Instead, researchers genetically encode desired mutations, by making mutated copies of a plasmid that contains the inverse pericam DNA sequence. In addition to non-mutagenic amplification of a specific piece of DNA, synthetic primers can be used to incorporate desired mutations into the DNA. Primer design for site-directed mutagenesis is quite straightforward: the forward primer introduces a mutation into the coding strand. Both non-mutagenic and mutagenic amplification require cycles of DNA melting, annealing, and extension.
Remember from Day 1 that primers used in PCR amplification must meet several design criteria in order to ensure specificity and efficiency. Consider the following design guidelines for mutagenesis primers and think about how these differ from the guidelines for non-mutagenic amplification:
- The desired mutation (1-2 bp) must be present in the middle of the forward primer.
- The forward and reverse primers should 'face' away from the mutation and be 'back-to-back' when annealed to the template.
- The primers should be 25-45 bp long.
- A G/C content of > 40% is desired.
- Both primers should terminate in at least one G or C base.
- The melting temperature should exceed 78°C, according to:
- Tm = 81.5 + 0.41 (%GC) – 675/N - %mismatch
- where N is primer length, and the two percentages should be integers.
To demonstrate primer design, the illustration below uses S101L, which is an uninteresting mutation but is a straightforward teaching example.
Residue 101 of calmodulin is serine, encoded by the AGC codon. This is residue 379 with respect to the entire inverse pericam construct, and we can find it and some flanking code in the DNA sequence from Part 2:
361 (5') GAG GAA ATC CGA GAA GCA TTC CGT GTT TTT GAC AAG GAT GGG AAC GGC TAC ATC AGC GCT (3')
381 (5') GCT CAG TTA CGT CAC GTC ATG ACA AAC CTC GGG GAG AAG TTA ACA GAT GAA GAA GTT GAT (3')
To change from serine to leucine, one might choose TTA, TTG, or CTN (wherer N = T, A, G, or C). Because CTC requires only two mutations (rather than three as for the other options), we choose this codon.
Now we must keep >10 bp of sequence on each side in a way that meets all our requirements. To quickly find G/C content and see secondary structures, look at the IDT website. (Note that the Tm listed at this site is not one that is relevant for mutagenesis.)
Ultimately, your forward primer might look like the following, which has a Tm of almost 81°C, and a G/C content of ~58%.
5’ GG AAC GGC TAC ATC CTC GCT GCT CAG TTA CGT CAC G 3'
The reverse primer is the inverse complement of a sequence just preceding the forward primer in the IPC gene. The forward and reverse primers are set up back-to-back.
Lucky for us, NEB has a tool that can design our mutagenic primers.
- Go to the NEBaseChanger site and click 'Please enter a new sequence to begin.'
- A new window will open. Copy and paste the wild-type IPC sequence.
- Confirm that the 'Substitution' option is selected.
- Highlight the basepairs you want to mutate using by scrolling through the sequence, or you can search the sequence by typing the basepairs into the 'Find' box.
- Type the new DNA sequence (the basepair(s) you want your forward mutagenic primer to incorporate into the IPC sequence) in the 'Desired Sequence' box.
- Under the Result header, a diagram showing where your primers will anneal is provided.
- Under the Required Primers header, the sequences for your forward primer and reverse primer are shown with the characteristics for each.
- Screen capture the information provided in the Result and Required Primers sections.
- Embed the images in your notebook.
- Print the screen capture and submit it to the teaching faculty before you leave today. In addition, record your primer sequences in the table on the Discussion page.
- It is very important that you submit your primer sequences before you leave! The teaching faculty will order your primers from IDT DNA tonight to ensure they arrive by your next class.
- Use the guidelines above to examine the mutagenesis primers designed by NEBaseChanger. Include your thoughts in your notebook.
- Do NOT alter the primers provided by NEB.
Part 2: Primer preparation
While you were away the sequences for the mutagenic primers you designed were submitted to Integrated DNA Technologies (IDT). IDT synthesized the primers then lyophilized (dried) them to a powder. Follow the steps below to resuspend your primers.
- Centrifuge the tubes containing your lyophilized primers for 1 min.
- Calculate the amount of water needed for each primer (forward and reverse, separately) to give a concentration of 100 μM.
- Resuspend each primer stock in the appropriate volume of sterile water, vortex, and centrifuge.
- Now prepare a dilution from your archival stock. Prepare 100 μL of a solution that has both the forward and reverse primers, each primer at 10 μM.
- Try the calculation on your own first. If you get stuck, ask the teaching faculty for help.
- Be sure to change tips between primers!
- Return the rest of your primer stocks, plus your primer specification sheets, to the front bench.
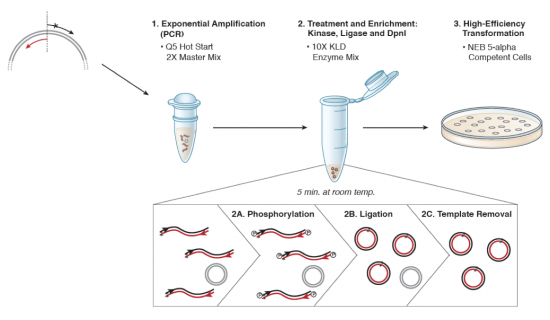
Part 3: Site-directed mutagenesis
We will be using the Q5 Site Directed Mutagenesis Kit from NEB to perform your site-directed mutagenesis reactions. Each group will set up one reaction, for your X#Z mutation. Meanwhile, the teaching faculty will set up a single positive control reaction, to ensure that all the reagents are working properly. You should work quickly but carefully, and keep your tube in a chilled container at all times. Please return shared reagents to the ice bucket(s) from which you took them as soon as you are done with each one.
- Get a PCR tube and label the top with your mutation and lab section (write small!).
- Add 10.25 μL of nuclease-free water.
- Add 1.25 μL of your mutagenesis primer mix (each primer should be at a concentration of 10 μM).
- Add 1 μL of IPC template DNA (concentration of 25 ng/μL).
- Lastly, use a filter tip to add 12.5 μL of Q5 Hot Start High-Fidelity 2X Master Mix - containing buffer, dNTPs, and polymerase - to your tube.
- Once all groups are ready, we will begin the thermocycler, under the following conditions:
| Segment | Cycles | Temperature | Time |
|---|---|---|---|
| Initial denaturation | 1 | 98 °C | 30 s |
| Amplification | 25 | 98 °C | 10 s |
| 55 °C | 30 s | ||
| 72 °C | 2 min | ||
| Final extension | 1 | 72 °C | 2 min |
| Hold | 1 | 4 °C | indefinite |
- After the cycling is completed, the teaching faculty will complete the KLD reaction (which stands for "kinase, ligase, DnpI") using 1 μL of your amplification product, 5 μL 2X KLD Reaction Buffer, 1 μL KLD Enzyme Mix, and 3 μL nuclease-free water. The reactions will be incubated for 5 min at room temperature.
- The teaching faculty will then use 5 μL of the KLD reaction product to complete a transformation into an E. coli strain (NEB 5α cells of genotype fhuA2 Δ(argF-lacZ)U169 phoA glnV44 Φ80 Δ(lacZ)M15 gyrA96 recA1 relA1 endA1 thi-1 hsdR17) that will amplify the plasmid such that you are able to confirm the appropriate mutation was incorporated. The transformation procedure will be as follows:
- Add 5 μL of KLD mix to 50 μL of chemically-competent NEB 5α.
- Incubate on ice for 30 min.
- Heat shock at 42 °C for 30 s.
- Incubate on ice for 5 min.
- Add 950 μL SOC and gently shake at 37 °C for 1 h.
- Spread 50 μL onto LB+Amp plate and incubate overnight at 37 °C.
Reagent list
- Q5 Site Directed Mutagenesis Kit from NEB
- Q5 Hot Start High-Fidelity 2X Master Mix
- Propriety mix of Q5 Hot Start High-Fidelity DNA Polymerase, buffer, dNTPs, and Mg2+.
- 2X KLD Reaction Buffer
- 10X KLD Enzyme Mix
- Proprietary mix of kinase, ligase, and DpnI enzymes.
- Q5 Hot Start High-Fidelity 2X Master Mix
- The SOC medium contains 2% tryptone, 0.5% yeast extract, 10 mM NaCl, 2.5 mM KCl, 10 mM MgCl2, 10 mM MgSO4, and 20 mM glucose.
- LB+Amp plates
- The Luria-Bertani (LB) broth contains 1% tryptone, 0.5% yeast extract, and 1% NaCl
- Plates prepared by adding 1.5% agar and 100 μg/mL ampicillin to LB