Procedure: Diffusion in biological gels
| |
This procedure uses sensitive biological samples. Wear gloves to avoid contamination. |
Summary
You are going to test the diffusion behavior of three different particle types in matrigel. The three particle species are:
a) 1 µm liposomes, positively charged (prepared for you in 1:200 dilution from of stock)
b) 1 µm liposomes, neutral (prepared for you in 1:200 dilution from of stock)
c) 500 nm polystyrene beads, positively charged (prepared for you in 1:50 dilution from of stock)
Dilutions of each of these analytes are prepared for you in tubes and will be kept on ice any day someone says they are going to do the particle tracking. Let us know as soon as you arrive for this experiment; we will then start to defrost a matrigel aliquot for you.
Part I: Making a sample Chamber
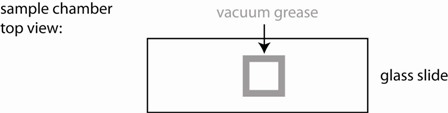
1) Use the vacuum grease filled syringe to place an open ~ 15x15 mm square on the middle of a glass slide. Make sure it is small enough so that it can be fully covered with a small cover slip. Ensure that the linings are continuous (see figure), otherwise your sample chamber will leak and you will have lots of drift.
1a) You will need to hold on to the base of the tip on the syringe, as it will tend to unscrew. If it does, simply screw it back in and hold on tigher.
2) Label the glass slide now to keep track of your sample. Be precise because your samples may last a few days if you keep them wrapped in foil. Include your group number!
3) Once your sample is prepared (see Part II below), you will place ~30 µl of that sample into the vacuum grease well of the sample chamber you crafted above. You may test this part of the procedure (once or twice only, please) using straight DMEM. Do not use the particles until your are making your sample in Part II.
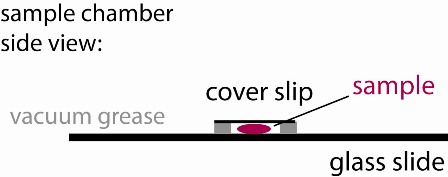
4) Cover the well with a small cover slip to create a sandwich (see figure). Press only enough to make contact with the sample and provide a continuous seal around the perimeter of your well. A bubble may remain inside the well. That is OK as long as it is stable. Try to keep the cover slip parallel to the slide. If some fluid leaks out, that is usually OK, just gently slide the cover slip around laterally to better ensure a continuous seal. If too much fluid has leaked, you may have a bad seal. To check, look around the perimeter at a glancing angle, using the bench ligths for illuination. If this does not work, ask a TA or Instructor to help you.
| |
It is important that your sample is not leaking out. This will cause flow that will complicate your tracking measurements. |
Part II: preparing the gel/particle mixtures
1) Defrost a 75 µl aliquot of matrigel on ice. (Your TA or Instructor will have an aliquot on ice if you have notified us in advance. Otherwise, you will have to wait approximately 45 minutes for the gel to slowly defrost, on ice, so notifiy us as soon as you arrive to the lab. The gels cannot be quickly defrosted, as this may cause gelation around the edges of the tube. And they should not be refrozen, as this may alter the experiment.)
2) Once the gel has thawed, place a different analyte into each of three test tubes, 1 µl each: use neutral liposomes in the first, positively charged liposomes in the second, and the polystyrene particles in the third.
3) Label your test tubes accordingly to keep track of your sample mixtures! Include your group number on the tubes.
| |
Keep all your aliquots on ice. |
4) Now place 25 µl DMEM in the two tubes containing the liposomes, and place 25 µl of your group's salt solution into the third tube with the nanoparticles. Mix by gentle pipetting under the surface, avoid creating bubbles! (Be aware, once you add the gel, bubbles will form easily, so mix even more carefully then, and do not "push out" the last of the gel solution. That will usually create many bubbles.)
NEVER vortex the gel solution!
5) Finally, distribute 25 µl matrigel into each of your three tubes.
| |
Keep all your aliquots on ice. |
You should now have three aliquots containing 51 µl of a gel/particle mixture each. Again, make sure they are labeled.
6) Now you are ready to fill each of your sample chambers. With very gentle pipetting, mix one of your tubes and fill the appropriate sample chamber with about 30 µl of fluid. Repeat for the other tubes/chambers. Of course remember to use a different tip for each. Once all chambers are full, place the cover slips and finish the chamber prep procedure described above.
7) Now place all of your filled sample chambers for 20 minutes in the incubator at 37°C to induce gelation.
8) Finally, remove your samples from the oven and place them on your vibration table to cool for maybe a minute, covering them at all times with foil or a box top to keep them from bleaching.
9) Your hydrogel samples are now ready for microscopy.