Assignment 3, Part 1: visualizing actin with fluorescence contrast
This is Part 1 of Assignment 3.
|
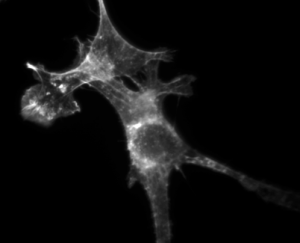
NIH3T3 mouse fibroblast cells. Actin stained with Alexa Fluor 568 Phalloidin. |
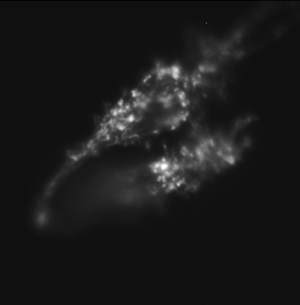
Same cell type incubated with cytochalasin D, an inhibitor of actin polymerization. |
|---|
Overview
In this part of the lab, you will use fluorescence microscopy to investigate the effect of a toxin called cytochalasin D on the actin cytoskeleton. Cytochalasin D is an actin polymerization inhibitor. Because it disrupts one of the main structural components of cells, cytochalasin D causes marked changes in shape and mechanical properties. In this part of the lab, you will make images of NIH 3T3 cells that have been exposed to cytochalasin D and cells that have not been exposed. In the next couple of assignments, you will investigate more deeply the effect that disrupting the actin cytoskeleton has on the mechanical properties of cells.
But wait... is it even possible to take a picture of actin? Remember the five requirements to make an image of something:
- Optical access? Check. We will grow the cells in a single layer in a glass-bottom culture dish. The cells are nearly transparent. Optical access is easy.
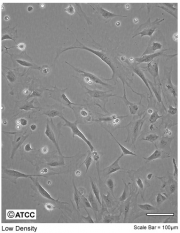
- Contrast? Nope. As you might be able to tell from the phase contrast image of 3T3 cells, actin does not generate contrast.
- Specificity? Nope. Without contrast, it's hard to have specificity.
- Resolution? Sort of. Actin stress fibers can have a long dimension on the order of microns, which is easy to resolve with a 0.65 NA objective. But they are crazy thin — about 7 nm. So even using the shortest possible wavelength of light, there would be no way to tell whether a line in the image represents a single fiber or many fibers close together.
- Noise margin? Without contrast, there is no noise margin.
Brightfield or phase contrast microscopy satisfies one requirement out of five. Clearly, we are going to have to take another approach in order to make the actin visible. Fluorescence to the rescue! There are (at least) three methods using fluorescence that will allow us to make an image of the actin cytoskeleton with high contrast and specificity:
- use genetic manipulations to modify the actin protein so that it fluoresces,
- use an antibody conjugated to a fluorescent dye, or
- use another non-antibody chemical compound that has high affinity for actin conjugated to a fluorescent dye.
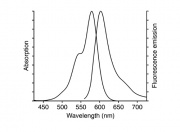
Each of the three approaches has advantages and drawbacks. All three approaches have been used in research studies, but we are going to use the third one. This will require fixing the cells before permeabilizing and staining them. For this to work, we need a chemical that binds to filamentous actin (F-actin) and a nice, bright fluorescent dye. Phalloidin is a mushroom toxin that binds to polymerized (F-actin) much more tightly than to actin monomers (G-actin). It has the specificity we are looking for. It is possible to connect phalloidin molecule to a fluorescent dye molecule. But will it work? I won't keep you in suspense any longer. Phalloidin retains its high affinity for F-actin even when it is conjugated to a dye! Victory! Even better, you can buy pre-made Alexa Fluor 568 Phalloidin from Invitrogen. Alexa Fluor 568 has high quantum yield and is reasonable resistant to bleaching. The images will be spectacular. I bet you can hardly wait.
Fixing and staining procedure
In outline, the procedure is:
- Treat one sample with Cyto D. Leave the other one untreated.
- Fix the cells with formaldehyde.
- Permeabilize the cells with Triton X 100
- Incubate with bovine serum albumen (BSA) to block nonspecific binding sites.
- Stain with Alexa Fluor 568 Phalloidin.
A dictionary definition of fix is: "to make firm, stable, or stationary." Formaldehyde fixation develops covalent cross-links between molecules in the cell — essentially gluing the bits together to from an insoluble meshwork. Cells must be permeabilized prior to staining so that the stain molecules can get inside and do their job. Permeabilization uses a detergent to partially dissolve the cell membrane. Without prior fixation, structures in cells would fall apart after permeabilization and diffuse away before you could stain them. Thus, cells must be fixed before immunostaining. Incubating with BSA prior to staining reduces dye binding to nonspecific sites.
Notes
- You will receive two plates of cells. One should be treated with cyto D and the other should be left untreated. The fixing and staining procedure is the same for both dishes. Read through the entire protocol before you begin. As noted in the protocol, some steps can be done in parallel.
- Avoid avoid shocking live cells with "cold" solutions (20 °C). Any reagents you add to live cells should be pre-warmed to 37 °C. in the water bath. After the formaldehyde step, the cells are no longer alive.
- Pipette carefully. That means slowly. Never pipette directly on to the cells. Cells wash off easily.
- Image your samples right after you stain them for best results
- If you can't image your fixed and stained samples right away, seal the dishes with parafilm, cover in foil, and label with your lab group's name. Store in 4 °C refrigerator. Remember to leave the PBS in the dish. They will last for a week or so, but time certainly does not make them better.
Equipment
- Personal protection equipment (PPE): gloves, lab coat, goggles when pipetting
- Pipettes and tips
- 37 °C water bath
- Waste disposal containers
Materials and reagents
- 2 glass bottom, 35 mm culture dishes with ~60% confluent 3T3 cells
- 0.8 ml 3.7 % formaldehyde
- ~45 ml phosphate buffered saline (PBS) at pH 7.4
- 1 aliquot (1 ml) cytochalasin D
- 3 ml Triton X-100
- 3 ml BSA
- 1 aliquot(400 μL) Alexa Fluor 568 Phalloidin (pre-mixed in methanol/PBS)
- Aluminum foil
Procedure
| |
Wear gloves and lab coats when you are handling biological samples. |
- Assemble materials and reagents. Wrap formaldehyde in foil to protect from light. Pre-warm formaldehyde, PBS, and cyto D in 37°C water bath.
- Label the samples "1/Cyto D" and "2/Control"
The steps below apply to DISH 1 ONLY — the one you will treat with cyto D.
- Aspirate the medium from DISH 1
- Wash 2X with 2 mL of pre-warmed PBS. Be gentle.
- Add 1 mL of pre-warmed 10 μM CytoD solution and incubate at 37 °C for 15 minutes. Watch the time closely. Over-treating will make all of the cells go away and you will be sad.
- Wash 2X with PBS and aspirate. (Be SUPER gentle. Treatment with cyto D makes the cells even more likely to wash off the coverslip.)
These steps apply to DISH #2 ONLY
- Aspirate the medium from DISH 2
- wash 2X with 2 mL of pre-warmed PBS and aspirate
These steps apply to BOTH dishes
- Pipet 400 μL of 3.7% formaldehyde solution ... as gently as you possibly can ... onto the cells in the central glass region of each dish.
- Let stand for 10 minutes at room temperature.
- Wash each dish 3X with 1.5 mL PBS
- Add 1.5 mL 0.1% Triton X-100
- Let stand for 3-5 minutes.
- Wash 2X with 1.5 mL PBS.
- Add 1.5 mL 1% BSA in PBS
- Let stand for 20 minutes.
- Wash 2X with 1.5 ml PBS.
- Add 200 μL of Alexa Fluor 568 phalloidin solution. Carefully pipet the solution just onto the center of the dish,
- Cover with aluminum foil
- Let stand for 45 minutes at room temperature.
- Wash 3X with PBS. Do not aspirate the last wash (leave the PBS in) so the cells don't dry out.
Fluorescence imaging
In this part of the lab, you will take images of your dishes of cells, and correct them for nonuniform illumination. In order to do the correction, you will need a reference image and dark images in addition to the image of the sample. See this page for more information about flat-field correction.
- Reference Images: Take the reference image as close as possible in time to the sample images. Don't make any adjustments to your microscope between capturing the reference image and the sample image. For example, every time you bump the camera or re-align the LED illumination path, you will change the illumination profile and you must take a new reference. Adjusting the camera exposure and gain between recording the reference and sample images is okay.
- Dark images: Each time you record an image (reference or sample), make sure to take a corresponding dark image using the EXACT SAME camera settings (i.e. use the exact same Exposure Time and Gain settings you had chosen for your reference/sample image). This is the only valid way to subtract the correct dark value from your reference/sample image.
- Saving: Remember to save your images in a format that preserves all 12 bits. We recommend using the MATLAB save command to save data in a .mat file so you can reanalyze it later if necessary. You could also save in an image file format with imwrite. Convert the image to 16-bit, unsigned integer format with the correct range before saving. Go back and read this page if you need to refresh your memory.
Tips
Visualizing the actin cytoskeleton under your 20.309 microscope will require mad skills. Since actin filaments and stress fibers are nanometer-scale objects, they are much dimmer than fluorescent beads or the dye solution - care must be taken to get good images of the cytoskeleton.
- Use a reference slide to check that your epi-illuminator path is still optimally aligned. The cells are very dim; you won't do yourself any favors if you are throwing away light.
- You may need to cover the microscope with a box or turn off the overhead lights to reduce room light contamination.
- As soon as you expose the sample to high power LED light, the fluorescent dye will start to photobleach. You may find it helpful to use your brightfield microscope to find your cells before turning on your green LED illumination. Make sure the lab power supply is set to "INDEP." if you want to use Ch1 and Ch2 independently.
- Once you've found cells in the brightfield, turn off the trans-illumination (red) LED and turn on the epi-illuminator. You will want to turn up the intensity of your LED, which can be achieved by increasing the current limit on the power supply, but never exceed 1A of current!
- Fine tune the focus using a high gain/low exposure setting. (This setting will give you poor images with a lot of noise, but it's hard to focus the sample with a long exposure time.) Using the stretch contrast' tick box in the acquisition software may help to increase the image contrast on screen and make it easier to focus the image.
- Finally, adjust the gain (to zero) and increase the exposure of the camera to get the best picture in terms of the signal-to-noise ratio.
- For each microscopy sample, remember to check the exposure time, and take a corresponding dark image. Have someone record the camera settings used for each image.
Take images
- Record a reference image
- Using a low intensity for your epi-illumination LED (i.e. a small current), take an image of the reference slide with the 40X objective.
- Use the histogram in the UsefulImageAcquisitionTool to be certain that the image is exposed correctly
- Turn off the LED and record a dark image (without changing any camera settings!)
- Using up to 1A of epi-illuminator LED current, record images of:
- Fixed and stained untreated cells and a corresponding dark image
- Fixed and stained cells treated with CytoD and a corresponding dark image
- Again, be certain that the images are exposed correctly
Flat field correction
Perform flat-field correction on the images.
- Divide the image by a normalized version of your reference image minus the dark image (see this page for more detail).
| |
Turn in a figure with images of the the stained cell samples with and without Cyto-D.
|
Supplementary information
Cell culture protocol
Here is the procedure used to produce your samples:
NIH 3T3 were cultured at 37°C in 5% CO2 in standard T75 flasks in DMEM++ (Dulbecco's Modified Eagle Medium [Invitrogen] supplemented with 10% fetal bovine serum [FBS - Invitrogen), 1% penicillin-streptomycin [Invitrogen], 1% non-essential amino acids, and 1% glutamine.) The day prior to the fluorescence imaging, cells were plated on 35 mm glass-bottom cell culture dishes (MatTek).
- Overview
- Part 1: Visualizing actin
- Part 2: Problems
Back to 20.309 Main Page