20.109(S21):M2D4
Contents
Introduction
Today is the culminating day of Module 2 and hopefully you will identify 'hits' from your SMM screen! Though you may be able to qualitatively visualize the spots that appear to emit more fluorescence, it is important to complete quantitative analysis that supports your observations. During our previous laboratory session, you learned that a microarrayer was used to read the fluorescence signals emitted from the surface of the SMM slide at two excitation wavelengths. As noted previously, the 532 nm wavelength was used to excite fluorescein, which was printed in an 'X' pattern to assist with alignment. The 635 nm wavelength was used to excite Alexa Fluor 647; which would be associated with TDP43-RRM12 bound to a small molecule on the slide. A hit denotes a spot on the slide that emits a red fluorescence signal significantly higher than the background fluorescence level. In terms of protein binding, a hit denotes that the TDP43-RRM12 protein is bound to a small molecule and is therefore localized to a specific position on the slide. You will analyze the fluorescence signal collected by the microarray scanner using a value termed the robust z-score.
The robust z-score differentiates signal from noise by providing a value that represents the intensity of a signal above background. In the case of the SMM experiment, the intensity of a fluorescent signal above the background fluorescence is calculated. To do this the fluorescence emitted across the entire slide is grouped to define the Median Absolute Deviation (MAD), which is is a measure of the variability of a univariate dataset. Though beyond the scope of this class, the equation for calculating the robust z-score assigns a value for how much more intense the fluroescent signal at a spot is over background. The higher the value, the more different the signal from background.
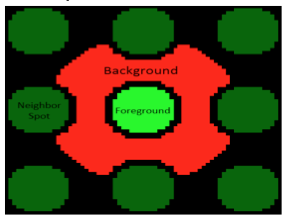
When the SMM slides were imaged, the microarrayer also produced a GAL file, or GenePix Array List. The GAL file contains information about where each spot was printed, and what compound was printed there. However, the relationship between the GAL file and the actual contact of the print head is very imprecise. Instead, we will use the fluorescein guide spots to align the array in the GAL file to the true print location for each pin. Following the alignment, we will compare the fluorescence at 635 nm within the deposition region of each spot (foreground) to the fluorescence immediately outside of this region, where nothing was printed (background) as illustrated in the image to the left. These values will be used to calculate the robust z-score. From the robust z-score, you can determine the associated probability that the observed fluorescence occurred by chance, and if this probability is sufficiently low, we call the small molecule a hit.
Protocols
Part 1: Identify hits from SMM results
To complete the data analysis steps outlined, you will use a Jupyter notebook generated by Rob Wilson, a researcher in the Koehler Laboratory, with additional edits by Caleb Perez. Though the code is provided to you, it is important to critically think through the data analysis steps!
- If you do not have Anaconda installed on your computer, please do so. Anaconda Installation Site.
- Download the .yml file to create your environment from Dropbox link here: yml file
- Download SMM files from Dropbox link here: SMM Files. Be sure to download the entire contents of the folder.
- Open the Anaconda navigator and choose “Environments” from the left tab
- Select “Import” on the bottom tab to upload the .yml file
- Name: SMM
- The true name of your environment is 20109_SMM, and it will automatically change to that once the yml file is uploaded.
- Name: SMM
- The .yml should automatically begin to populate the new environment with widgets necessary to correctly run the SMM analysis.
- This will take a few minutes. If you prefer to build your environment manually, please see instructions linked here Create SMM environment in Conda
- Return to the Home section of Anaconda navigator and ensure that that the 20109_SMM environment is selected. You should be able to see Application on 20109_SMM on the top of the window.
- Locate the Jupyter Notebook panel and click on the gear at the top right corner.
- Choose “Install specific version” and select 6.1.1
- Once the Jupyter Notebook is updated, launch the notebook.
- The Jupyter notebook directory should open in a new window.
- To select the Jupyter notebook location, select the location you have downloaded the SMM files, open the '20200221_TDP43-Screen' directory, then select the notebook '20109_S21_SMM_Analysis.ipynb'.
- The notebook should look like the image below:
- The text provides details on what the code below (in boxes) is accomplishing. As you work through the data analysis, make notes in your laboratory notebook regarding the steps. Specifically, include the purpose for each step. You will also generate several figures as you complete the analysis, these should also be included in your laboratory notebook with some description of what is shown.
In your laboratory notebook, complete the following:
- For each step in the code used to analyze the SMM data, provide a brief description of the purpose (ie what is the code doing?).
- If a figure was generated at the step, include it!
- Attach a list of the 'hits' you identified.
Part 2: Visually evaluate chemical structures of positive hits
Proteins and small molecules interact in a process referred to as molecular recognition. In molecular recognition, the non-covalent complexes that form are defined by two characteristics: specificity and affinity.
- Specificity distinguishes a specific binding partner from the milieu of potential binding partners in complex environments.
- Affinity dictates the likelihood of binding based on the concentration of a specific binding partner in a milieu of potential binding partners such that high affinity partners at low concentrations are not outcompeted by low affinity partners and high concentrations.
In cells, proteins are critical macromolecules that perform numerous roles related to structure, mechanics, metabolism, and signaling. In these roles, proteins do not work in a vacuum; rather the biological roles of proteins are dependent on direct physical interactions with other molecules. Though our focus is on small molecule binders, proteins also interact with other proteins, nucleic acids, oxygen, and metals. In all cases, the interactions are characterized by protein-ligand-solvent binding kinetics.
Though beyond the scope of this module, protein-ligand-solvent binding kinetics are defined as a thermodynamic system composed of solute (proteins and binders) and solvent (liquid that contains the proteins and binders). In that protein-small molecule complexes result in heat transfer, the driving forces that promote these interactions are due to energy exchanges that are characterized by Gibbs free energy (ΔG). ΔG measures the capacity of a thermodynamic system to do maximum or reversible work at constant temperature and pressure. When at equilibrium with constant temperature and pressure, protein-small molecule binding occurs when the change in ΔG is negative. The magnitude of ΔG provides insight into the stability of a protein-small molecule complex.
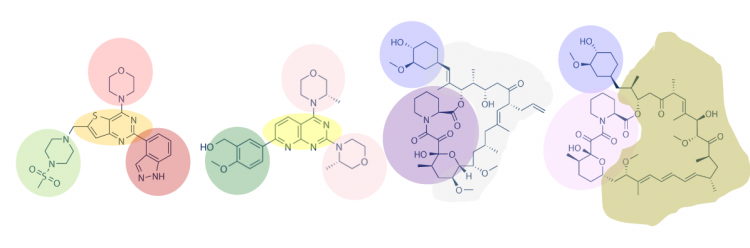
Another method for assessing protein-small molecule binding is to visually inspect known small molecule binders for common features / structures. Though you may not realize it, you did this on M2D1! Remember the information regarding lactose and IPTG? In both of these examples, the molecules share a feature that enables binding to the same target (LacI for lactose and IPTG). Your goal for today is to carefully examine the hits identified by the class and identify any common features / structures. As in the image below, it is possible that multiple features will be present within the same small molecule.
With your partner, review the hits identified in the SMM. It may be easier to copy / paste the small molecule images into a powerpoint file so you can readily see all of the structures. Also, it may be helpful to use a color-coding system (like the one in the image provided in the Introduction) to highlight features / structures that are common to the small molecules that putatively bind TDP43-RRM12.
These online resources may be helpful to learning more about the hits identified in the SMM:
- Cloud version of ChemDraw here.
- Copy and paste the small molecule smiles into the work space to get a chemical structure
- Platform to transform the smiles information into a PubChem ID here.
- Copy and paste the smiles into the input ID search to determine the ID number.
- PubChem database of chemical information here.
- Includes small molecule molecular weight and other useful information.
In your laboratory notebook, complete the following:
- How many features did you identify that are present in two or more of the small molecules that putatively bind TDP43-RRM12? Are there more or less than you expected?
- Is there a feature present in all of the identified small molecules? What might this suggest about the binding site(s) and / or binding ability of TDP43-RRM12?
- Can you assign the identified small molecules to sub-groups based on the common features that are present?
- What might the different features represent? More specifically, consider whether each subgroup has a unique binding site on the target protein or if each subgroup represents different solutions for interacting with the same binding site. They can propose studies (along with underlying rationale) that would help them distinguish these possibilities.
- How might you make modifications to the small molecules / features to probe binding? As a hint, consider how different functional groups could be positioned at a given site without altering qualitative binding in the SMM assay to translate that into some testable ideas (e.g. quantitative binding properties may be occurring that are functionally relevant, but not discernible by SMM assays; or such a site is not critical for binding and may allow for modifications that confer beneficial properties of the compound).
Part 3: Design experiments to test putative small molecule binders
Using the ideas discussed with your partner for Part 2, design the experiments that will follow your research. Remember, that the next steps to your project should build upon the work you completed.
In your laboratory notebook, complete the following:
- Consider aspects of the experimental approach that could be improved.
- What steps might you do differently to improve the results?
- Consider two potential next step experiments that you might perform to test putative small molecule binders identified from the SMM screen. For each experiment:
- What specifically will the experiment test?
- How will this experiment bolster the results from the current research project?
- Is the experiment a next step? Does the experimental approach make assumptions beyond the data acquired from the SMM screen?
- Include some information regarding a method that could be used to complete the experiment. What controls should be included?
- Consider the implications of your research.
- How does your research address the knowledge gap / research question? This should tie back to the information provided in the Introduction of your Research article.
- How does your research advance the field of neurodegenerative diseases? Don't overreach, but do state what is now known because of your work.
Next day: Learn best practices for mammalian cell culture