20.109(S21):M1D6
Contents
Introduction: Characterize clone ligand binding using flow cytometry
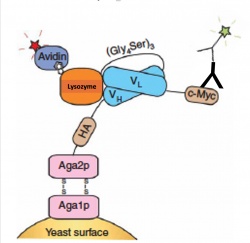
The goal of this module is to find a scFv clone with increased stability and affinity for our antigen lysozyme. Today we will carry out a quantitative binding experiment to measure the binding affinity via flow cytometry. Binding affinity is usually measured and reported as an equilibrium dissociation constant , denoted Kd. We will measure binding with a similar approach as our initial library screen. In our initial screen we had many different yeast clones(~1 million) expressing scFvs, and we incubated these yeast with one concentration of lysozyme. Today to calculate the equilibrium dissociation constant we will titrate a single scFv clone with varying concentrations of lysozyme. As we did on M1D2 we will use AlexaFluor488(green) labeled antibodies to mark the scFv on the surface of our yeast clone and AlexaFluor647(far red) labeled streptavidin to mark the presence of bound biotinylated lysozyme as seen in Figure 1.To accurately measure the binding affinity of your clone you will measure eight concentration of ligand, biotinylated lysozyme, with the same number of yeast expressing one clone. The concentration range should ideally span two orders of magnitude both above and below the estimated Kd of the clone being measured, but practical considerations of tube volumes or reagent usage may limit the ability to achieve this goal. The amount of time you incubate the ligand and scFV should be considered as we are measuring binding at equilibrium. Practically for most antibody-antigen interactions 30minutes is sufficient to reach equilibrium, but one to two hours is common practice. After equilibrium binding is met, the lysozyme/yeast mix should be kept on ice to minimize antigen dissociation.
To set up our flow cytometry gates we will also need to prepare control samples for each clone. We will prepare two negative control samples, the first is the yeast clone with no fluorophores or ligand added. The second negative control is the yeast clone with secondary only with no primary antibody or lysozyme. Finally the positive control is considered the titration condition with a high concentration of lysozyme that is likely to bind well and give a strong signal. This is the clone incubated with lysozyme at two orders of magnitude above the estimated Kd of the clone.
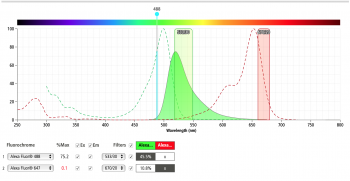
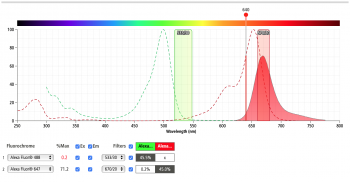
Prior to our experiment we can use the BD Spectrum Viewer to check the excitation and emission profiles of our fluorophores of choice on the analyzer we are using for our experiment. The figures below show the result of the excitation and emission of AlexaFluor488 (left) and AlexaFluor647 (right) on the Accuri C6 flow cytometer. Do you predict we will have significant signal strength on the Accuri C6 with our fluorophores of choice?
To start the analysis, the unstained negative control yeast population is gated on forward- and side-scatter channels to remove debris and aggregated cells. The secondary only controls are used to remove background fluorescence from fluorophores non-specifically sticking to the yeast clone. Then the positive control samples are used to establish the fluorescent signal from scFv expression(green) and lysozyme binding(red).
Protocols
Part 1: Prepare Titration Lysozyme with scFv Clone on Yeast
Motivation: To determine the equilibrium binding constant of our mutant scFv clone we will incubate a single yeast clone with eight different concentrations of lysozyme and measure binding via fluorescence intensity by flow cytometry.
Prior to this session, the instructors transformed your clone plasmids back into yeast and grew transformed colonies on SDCAA selective plates. Next, they picked an individual colony to grown in SDCAA media culture. After a day of growth, the culture was passaged (i.e. spun down and resuspended) into SGCAA media to induce expression of your scFv on the surface of the yeast via galactose induces Gal1 promoter.
Retrieve yeast culture labelled with your groups color from 20°C incubator
- Measure the optical density (OD) of your yeast on the spectrophotometer.
- Blank the spectrophotometer with >1000 uL of SGCAA media in a clear cuvette
- Add 900 uL of SGCAA media to a new cuvette. Add 100 uL of your yeast culture and mix well by pipetting up and down.
- Record the OD(@600nm) of your sample by taking a measurement using the spectrophotometer and multiplying by 10 (to account for the 10x dilution).
- Determine how many microliters of your sample are required to analyze 1x106 yeast cells.
- The conversion rate from OD to cells for yeast is: (107 yeast cells)/(OD x mL of culture)
- Thus, An optical density of 1 at 600nm equals 107 yeast cells per mL.
- Add 10 microcentrifuge tubes to your rack and label them 1-10 with tape matching your team’s color.
- Add the correct volume of culture (calculated above) such that 10^6 yeast are added to each tube.
- Add an additional 900 uL of PBSA to each centrifuge tube and pellet the cells for 2 minutes in microcentrifuge at 4000xg.
- Discard supernatant carefully with vacuum aspirator or with pipette (Do not touch or disturb the yeast pellet!). Wash with an additional 1 mL of PBSA, pellet, and remove supernatant. Repeat washing step once more.
Retrieve biotinylated lysozyme stock solution from instructors
- Use additional microcentrifuge tubes to prepare dilutions of lysozyme stock solution in PBSA
- Start by making a 2000 uL sample of 1000 nM biotinylated lysozyme. Then, calculate and prepare dilutions from that sample in additional microcentrifuge tubes (starting with 1500 uL of 316 nM solution, then 1500 uL of 100 nM solution, etc.)
- Add 1000 uL of correct concentration of lyzosyme solution to each labeled sample tube as directed below (Tubes 1-8). Carefully, resuspend the pelleted yeast. Add 1000 uL of PBSA (no lysozyme) to Tubes 9 and 10.
- Tube 1 = 1000 nM biotinylated lysozyme + 106 displaying yeast cells
- Tube 2 = 316 nM biotinylated lysozyme + 106 displaying yeast cells
- Tube 3 = 100 nM biotinylated lysozyme + 106 displaying yeast cells
- Tube 4 = 31.6 nM biotinylated lysozyme + 106 displaying yeast cells
- Tube 5 = 10 nM biotinylated lysozyme + 106 displaying yeast cells
- Tube 6 = 3.16 nM biotinylated lysozyme + 106 displaying yeast cells
- Tube 7 = 1 nM biotinylated lysozyme + 106 displaying yeast cells
- Tube 8 = 0.316 nM biotinylated lysozyme + 106 displaying yeast cells
- Tube 9 = 106 displaying yeast cells (secondary staining only) (no lysozyme)
- Tube 10 = 106 displaying yeast cells (no staining) (no lysozyme)
- Add primary staining to samples: Add 1 uL of the chicken antibody specific for the c-myc tag (GaCmyc mAb) to Tubes 1-8
- Incubate samples on nutator at 4°C overnight.
- After incubation, pellet cells at 4000xg for 2 minutes. Discard supernatant carefully with vacuum aspirator or with pipette (Do not touch or disturb the yeast pellet!). Wash with an additional 1 mL of PBSA, pellet, remove supernatant, and resuspend with 1 mL of PBSA.
- Add secondary staining to samples: Add 1 uL of goat polyclonal antibody mix labelled with Alexa Fluor 488 specific for chicken antibodies (GaC488) and 1 uL of streptavidin labelled with Alexa Fluor 647 (SAV647) to Tubes 1-9.
- Incubate samples on nutator at room temperature for 20-30 minutes.
- After incubation, pellet cells at 4000xg for 2 minutes. Discard supernatant carefully with vacuum aspirator or with pipette (Do not touch or disturb the yeast pellet!) and resuspend with 1 mL of PBSA.
- Place samples on ice until analysis.
In your laboratory notebook, complete the following:
- Calculate the volumes of lysozyme stock (or volume of lysozyme dilution) to add to each tube (Tube 1-8) to arrive at the final concentration listed.
- Remember the stock concentration of lysozyme is 130uM and the volume you should resuspend the yeast pellet in is 1000uL.
- You are resuspending the lysozyme in PBSA buffer.
Part 2: Accuri Flow Cytometry of Stained Yeast Samples
Motivation: To measure the fluorescent signal from scFv expression and lysozyme binding per cell we will analyze the cells by flow cytometry. This will give us a fluorescent intensity value associated with each yeast cell.
To prepare for analysis, the Accuri flow cytometer needs to be flushed with fluid to remove any contamination from previous samples. Your samples will then be filtered one by one into round bottom analyzer tubes and placed under the cytometer sample injection port (SIP). The machine will then suck up the sample, and produce the dot plots. The Accuri will record the data from each laser and filter for every cell. The gating that the instructor performs will simply help visualize the samples as we perform the experiment but all the data will be saved automatically for post-processing analysis.
For more details on using the BD Accuri C6 flow cytometer, please watch the video tutorial linked here: Flow Cytometry. The steps are detailed below so you can follow along!
- Turn machine on. MilliQ water (ultra-pure water) should be in a tube on the sample injection port (SIP).
- Visually check waste containers and buffers.
- Open BD AccuriC6 software on the computer.
- To clean the machine, place a round bottom tube with cleaning solution on the SIP and under Run settings, choose "Run with Limits" and set 5 minutes and click RUN.
- Once the 5min cleaning is complete, place an empty tube on the SIP and under Run settings, click the backflush button to rinse the system.
- Place a fresh tube with 2ml of MilliQ water and run for 2min.
- You are now ready to analyze your samples.
- Place one filter cap tube in a rack and take your unstained yeast sample from the ice bucket.
- Invert the yeast microcentrifuge tube once, take the P1000 pipet and take up the yeast sample. Place the pipet tip flush against the filter cap and push the yeast sample through the 35um mesh into the round bottom tube.
- Remove the filter cap off of the yeast sample, take the MilliQ water tube off of the SIP and place the tube with filtered yeast on the SIP.
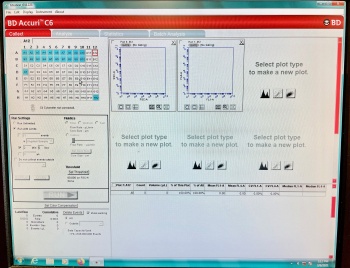
- Choose the analysis parameters in the software under Run settings. Run with limits: 10,000 events, instead of a time limit.
- Choose an analysis cell in the software with no events recorded and hit the RUN button.
- Unused cells are white (Example, C1) and cells with stored data are blue (Example, A1).
- We will detail gating and fluorescent analysis on M1D7.
- As the analysis proceeds, prepare your next yeast sample according to the steps above.
- Data is collected in individual cells in the software. Make sure to move the event record to a new cell between samples. Hit RUN each time you start a new sample analysis.
- Either name the cells with experimental details or write in your notes which samples are analyzed in each cell.
- Once all of the samples are analyzed, place a round bottom tube of Decon solution (10% bleach) on the SIP and run for 2min.
- The flow cytometer is now ready for the next user.
In your laboratory notebook, complete the following:
- What is the major difference between fluorescence activated cell sorting (FACS) and flow cytometry(flow)?
- For flow cytometry, we can set the gates after the whole experiment is done. Why is this the case?
- What are the similarities between the controls for FACS and flow? What are the differences?
- What are the similarities between the lasers and filters for the Aria II FACS and the Accuri C6 flow? What are the differences? check BD Spectrum Viewer for details. Choose Alexa Fluor 488 and Alexa Fluor 647 from the Fluorochrome dropdown. Change the laser excitation at the top of the page. The best choice for filters will auto-populate.
- What is one major benefit of FACS over flow?
- What is one major benefit of flow over FACS?
Reagents list
- SDCAA (Minimal yeast media composed of salts, Difco yeast nitrogen base, dextrose, and casmino acids)
- SGCAA (Minimal yeast media composed of salts, Difco yeast nitrogen base, galactose, and casmino acids)
- PBSA (Phosphate-buffered-saline solution with 0.1% w/v Albumin)
- Biotinylated lysozyme stock solution (prepared by instructors, Sigma Aldrich)
Primary Staining Reagents:
- Chicken anti-cMyc Ab (1:1000, Gallus)
Secondary Staining Reagents:
- Streptavidin AlexaFluor 488 conjugate (1:1000, Life Technologies)
- Goat anti-chicken AlexaFluor647 Ab (1:1000, Life Technologies).
- BD Accuri C6 flow cytometer
Next day: Analyze ligand titration curves
Previous day: Analyze clone sequences and choose clone to characterize