20.109(S17):Module 1
Contents
Module 1
Lecturer: Angela Koehler
Instructors: Noreen Lyell, Leslie McClain and Maxine Jonas
TA: Rob Wilson
Lab manager: Hsinhwa Lee
Overview
Chemical probes are important research tools used to explore cellular processes and therapeutic targets. The use of high-throughput and unbiased strategies to identify small molecules that bind specific biomolecules, such as proteins, can provide insight on the structure or function of targets. Additionally, a small-molecule screen can identify new chemical probes for target proteins of interest.
The small-molecule microarray (SMM) is a high-throughput method that enables the detection of protein-ligand binding. Briefly, ligands are 'printed' onto a slide and incubated with purifed protein. Unbound protein is washed from the slide and bound protein is detected using a tag on the protein of interest. Because the location of every ligand on the slide is known, the detection of protein indicates that it is bound to the ligand at that location.
In your experiment, you will use SMMs to identify ligands that bind to FKBP12, a folding chaperone for proteins that contain proline residues in eukaryotes.
This module has been developed thanks to the generous time and thoughtful efforts of several Koehler Laboratory members, in particular Rob Wilson, Dr. Becky Leifer, and Shelby Doyle.
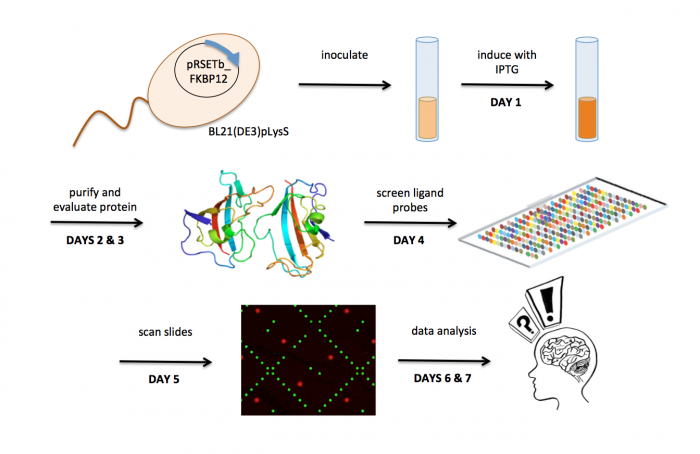
Lab links: day by day
M1D1: In silico cloning and induce protein expression
M1D2: Purify induced protein
M1D3: Evaluate purity and concentration of protein
M1D4: Screen ligand library for FKBP12 binders
M1D5: Scan slides to identify FKBP12 binders
M1D6: Complete data analysis
M1D7: Identify chemical structures common in FKBP12 binders
Data
See all M1 student data on the Discussion page.
Assignments
Data summary
Mini-presentation
References
- A method for the covalent capture and screening of diverse small molecules in a microarray format. Nature Protocols. 1:2344-2352.
- Recent discoveries and applications involving small-molecule microarrays. Chemical Biology. 18:21-28.