20.109(S15):Module 2
Contents
Module 2: System Engineering and Protein Foundations
Lecturer: Leona Samson
Instructors: |Shannon Hughes, |Noreen Lyell, and |Leslie McClain
Overview
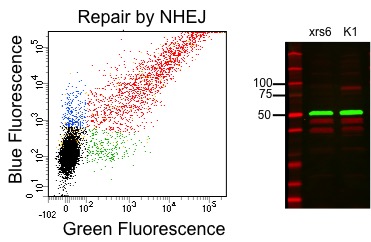
During the first module, you spent part of your time designing and testing a diagnostic tool for detecting a microorganism. Now you will have the opportunity to apply a research tool that detects a molecular process to address an interesting biological question. Specifically, you will investigate the importance of different proteins to a type of DNA repair called non-homologous end-joining, or NHEJ. Measuring DNA repair accurately and quantitatively can pave the way for certain cancer diagnostics and therapeutics.
This time the DNA engineering has been done for you: the plasmid-based research tool to measure NHEJ already exists, along with a novel version developed just for 20.109. In this second module you will instead take a systems level view, as you explore how different topologies of DNA damage are repaired by wild-type hamster (CHO) cells, CHO cells treated with a small molecule inhibitor of an NHEJ protein, and CHO cells that are natively deficient in an NHEJ protein. Along the way, you will gain additional skills in analyzing and communicating information related not only to DNA assays (as in Module 1), but also to protein and cell-level assays. The culminating experiment will utilize flow cytometry, an amazing and infinitely useful technique that measures the fluorescence of individual cells. To evaluate class-wide trends in the flow data, you will learn and use basic statistical tools. The toolkit you develop during the first two modules should leave you well poised to tackle the third and final module.
We thank Agi Stachowiak, a former 20.109 technical instructor, and Alex Chaim and Zac Nagel from the Samson lab for their development of this module, along with Marcus Parish for assistance with irradiation assays.
Lab Links
Day 1: Introduction to cell strains and plating
Day 2: Begin Western protein analysis and choose system conditions
Day 3: Complete Western and prepare damaged DNA
Note: spring break occurs between Days 3 and 4.
Day 4: Cell preparation for DNA repair system
Day 5: DNA repair assays
Day 6: Flow cytometry and paper discussion
Day 7: Data analysis
Assignments
NHEJ system research article
20.109 Blog summary