Difference between revisions of "20.109(F20):M1D2"
Noreen Lyell (Talk | contribs) (→Part 1: Treat cells for γH2AX assay) |
Noreen Lyell (Talk | contribs) (→Part 1: Treat cells for γH2AX assay) |
||
| Line 56: | Line 56: | ||
#*Stock concentration of H<sub>2</sub>O<sub>2</sub> is 10 M. | #*Stock concentration of H<sub>2</sub>O<sub>2</sub> is 10 M. | ||
#*Volume of 20 μM H<sub>2</sub>O<sub>2</sub> added to each well is 1 mL. | #*Volume of 20 μM H<sub>2</sub>O<sub>2</sub> added to each well is 1 mL. | ||
| + | |||
| + | '''As treatment steps''' | ||
| + | |||
| + | #Retrieve the 12-well plate with the H<sub>2</sub>O<sub>2</sub>-treated MCL-5 cells from the incubator and use a pipette to remove the media from the wells. | ||
| + | #Add 1 mL of fresh media or media + As to the appropriate wells and incubate at 37 °C for the designated amount of time. | ||
| + | #Following treatment, transfer media to the As waste container and immediately add 400 μL of 4% paraformaldehyde to fix the cells. | ||
| + | #Incubate at room temperature for 10 min. | ||
| + | #Collect the 4% paraformaldehyde in the correct waste stream using a P1000 pipet. | ||
| + | #Wash with 500 μL of 1X PBS. | ||
| + | #*Add 1X PBS then remove using a P1000 pipet. Collect the PBS in the correct waste stream. | ||
| + | #*Complete a total of 2 times. Leaving 1 mL of 1X PBS on the cells in the final wash. | ||
| + | #Leave all wells with 1 mL PBS, parafilm the sides and move the fixed plates into the 4 °C cooler. | ||
| + | |||
| + | |||
| − | |||
#During the MMS treatment, calculate the dilutions of EdU and As needed to have final concentrations of 5 μM and 80 μM, respectively. | #During the MMS treatment, calculate the dilutions of EdU and As needed to have final concentrations of 5 μM and 80 μM, respectively. | ||
#*EdU stock concentration is 10 mM and As stock concentration is 100 mM. | #*EdU stock concentration is 10 mM and As stock concentration is 100 mM. | ||
| Line 74: | Line 87: | ||
| Media | | Media | ||
| Final volume (1 mL / well) | | Final volume (1 mL / well) | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
===Part 2: Prepare CometChip=== | ===Part 2: Prepare CometChip=== | ||
Revision as of 15:22, 15 July 2020
Contents
Introduction
INCLUDE BER PATHWAY DESCRIPTION / PROJECT OVERVIEW
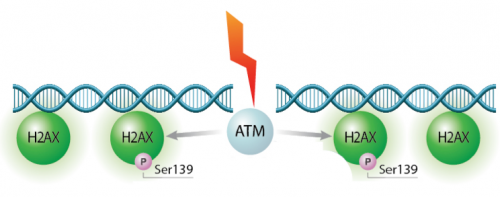
In eukaryotes, including humans, DNA is tightly wound around histone groups. H2AX is a member of the core group of histones that contributes to nucleosome formation and DNA structure. When a DNA double-strand break is introduced into the genome, the H2AX histones near the break are phosphorylated by the ATM kinase at residue Ser-139. Upon phosphorylation H2AX is referred to as gamma-H2AX. Given that only H2AX histones near the site of DNA damage are phosphorylated, γH2AX is a useful target when determining the abundance and location of double-strand breaks.
In your γH2AX assay, you will assess the effects of exposure to H2O2 +/- As.
Protocols
Part 1: Seed cells for γH2AX assay
To ensure the steps required for seeding cells are clear, the Instructor will provide a live demonstration of this process.
In your laboratory notebook, complete the following:
- Provide a written overview / description of the the procedure used to seed cells for the γH2AX assay (from the live demonstration).
- Calculate the volume of cell suspension added to each well for the γH2AX assay.
- The cell suspension is 1 M cells / mL and 25,000 cells were added into each well.
- For how long will the seeded cells incubate prior to treatment with H2O2 +/- As?
Part 1: Treat cells for γH2AX assay
A plate seeded according to the procedure demonstrated in Part 1 was used to treat cells using the H2O2 +/- As conditions detailed below.
Before you read through the protocols for the H2O2 +/- As treatments, it is important to consider what conditions will be assessed in this experiment.
With your laboratory partner, make a list of all of the conditions that will be tested. Use the following descriptions of the variables that are included in this experiment to assist you.
- Two concentrations of H2O2 will be tested: 0 μM and 20 μM
- Three concentrations of As will be tested: 0 μM, 1 μM, and 10 μM
- Each concentration of H2O2 will be tested with each concentration of As.
- Four timepoints will be used for each condition tested: 0 min, 15 min, 30 min, and 60 min.
In your laboratory notebook, include the following:
- A list or table of the conditions that will be used for the γH2AX assay.
- What conditions from your list or table are controls? For what does each condition control?
H2O2 treatment steps
Now that you know what conditions will be tested, the first step in the protocol is to treat the MCL-5 with H2O2.
- Retrieve the 12-well plate that was seeded with MCL-5 cells from the 37 °C incubator.
STEP FOR ADHERING CELLS
- After aspirating off the media in the wells, add 1 mL of fresh media or media + 20 μM H2O2 to the appropriate wells.
- Incubate at 37 °C for 1 hour.
In your laboratory notebook, complete the following calcultion:
- Calculate the dilution of H2O2 needed to have a final concentration of 20 μM.
- Stock concentration of H2O2 is 10 M.
- Volume of 20 μM H2O2 added to each well is 1 mL.
As treatment steps
- Retrieve the 12-well plate with the H2O2-treated MCL-5 cells from the incubator and use a pipette to remove the media from the wells.
- Add 1 mL of fresh media or media + As to the appropriate wells and incubate at 37 °C for the designated amount of time.
- Following treatment, transfer media to the As waste container and immediately add 400 μL of 4% paraformaldehyde to fix the cells.
- Incubate at room temperature for 10 min.
- Collect the 4% paraformaldehyde in the correct waste stream using a P1000 pipet.
- Wash with 500 μL of 1X PBS.
- Add 1X PBS then remove using a P1000 pipet. Collect the PBS in the correct waste stream.
- Complete a total of 2 times. Leaving 1 mL of 1X PBS on the cells in the final wash.
- Leave all wells with 1 mL PBS, parafilm the sides and move the fixed plates into the 4 °C cooler.
- During the MMS treatment, calculate the dilutions of EdU and As needed to have final concentrations of 5 μM and 80 μM, respectively.
- EdU stock concentration is 10 mM and As stock concentration is 100 mM.
- You will dilute in a final volume of 1 mL of media / well.
- Next, make a plan for the As treatment! Remember from prelab that the EdU will be applied to all of the cultured cells in your plate for 15 min. In the conditions with As treatment, the media containing both EdU and As must be replaced with media containing only As after 15 min. Conditions without As must be replaced with serum-free media after 15 min.
- To organize yourself, complete the table below to assist you in calculating the volume of each component needed to treat the duplicate wells in each condition. For example, in Condition #1 you should generate a mix that contains, 1 μL EdU and 2 mL media for a final volume of 2 mL (1 mL is added to each duplicate well...because 1 μL is a negligible volume in this instance there is no need to account for it in the total volume of the mix).
- Following the EdU incubation, the media in each well must be replaced with either media or media containing As. Create another table to assist you in preparing the mixes needed in this step of the procedure.
- Check all calculations with the teaching faculty prior to completing the remaining steps!
| Condition | EdU (final concentration = 5 μM) | As (final concentration = 80 μM) | Media | Final volume (1 mL / well)
Part 2: Prepare CometChip
Reagents listγH2AX
Next day: Use immunoflourescence staining to assess repair foci experiment |