Optical Microscopy Part 3: Resolution, Stability, and Particle Tracking
Overview
Congratulations on completing part 2. You made a functioning epifluorescence microscope. How cool is that?
In this part of the lab, you will measure the resolution of your microscope using tiny, fluorescent microspheres as sources. Next, you will quantify the stability of your microscope by tracking immobile beads. This will establish the position noise of your instrument as a baseline performance parameter. Then, you will characterize the diffusive motion of particles in different rheological environments (including live cells) by tracking fluorescent microspheres. Your microscope will act as a position detector.
Instructions
Measuring resolution
One of the most commonly used definitions of resolution is the distance between two point sources in the sample plane such that the peak of one source’s image falls on the first minimum of the other source’s image. The definition suggests a procedure for measuring resolution: make an image of a point source; measure the peak-to-trough distance in the image plane; and divide by the magnification. In this part of the lab, you will use a procedure inspired by this simple idea to estimate the resolution of your microscope. Instead of measuring the spot sizes with a ruler, you will use nonlinear regression on a digital image of point sources.
One practical problem with this method is that true point sources are difficult to come by. If you are using a telescope, stars are readily available to image and they are very good approximations of point sources. Since there is no natural microscopic sample that is equivalent to the night sky, microscopists have to construct something suitable. One of the most common facsimiles is a microscope slide sprinkled with tiny, fluorescent beads that have diameters in the range of 100-190 nm. These beads are small enough to be considered point sources. Unfortunately, beads small enough for this purpose are not very bright. Imaging them can be challenging. Your microscope must be very well aligned to get good results. To measure the spots, you will use nonlinear regression to fit a model curve to the
You will use image processing functions to locate the beads in your image and fit a Gaussian function to them. Gaussians are more amenable to nonlinear regression than Bessel functions, and they are a very good approximation. It is straightforward to convert the Gaussian parameters to Rayleigh resolution. See Converting Gaussian fit to Rayleigh resolution for a discussion of the conversion.
Procedure
- Make an image of a sample of 170 nm fluorescent beads with the 40X objective. (Several dozens to hundreds of PSF spheres should be captured in your image.)
- Use 12-bit mode on the camera and make sure to save the image in a format that preserves all 12 bits.
- Ensure that the image is exposed properly.
- Over-exposed images will give inaccurate results.
- Under-exposed images will be difficult to process and yield noisy results.
- This procedure is extremely sensitive to the focus adjustment.
- To minimize photobleaching, do not expose of the beads to the light source and longer than necessary.
- Be sure to save the image and the histogram for your lab report.
- Use image processing functions to locate non-overlapping, single beads in the image.
- Use nonlinear regression to fit a Gaussian to each bead image.
- Convert the Gaussian parameters to resolution.
- This page has example MATLAB code.
Stability of microscope for particle tracking
The accuracy of optical particle tracking may be limited by mechanical and optical phenomena. Vibration and drift are a source of additive noise. Shot noise and CCD readout noise in the image of a particle bring about uncertainty in the estimate of its centroid. Excessive vibration can frequently be corrected by improving the mechanical support structure of the instrument. Most stages can be locked to reduce drift. Shot noise is fundamental; however, its relative contribution to the total signal can be minimized by ensuring that the optical system is functioning at peak efficiency.
Before attempting to make measurements with particle tracking, it is essential to determine the performance characteristics of the instrument to be used. This can be accomplished by measuring a specimen with known characteristics. Perhaps the most foolproof choice is a sample with fixed particles. Any measured variation in the fixed sample is noise.
In order to analyze your data, you will need to write code in Matlab. It is helpful to make sure your code is working before you collect data. Problem Set 2 will help you with this. You may download this set of Matlab files to help you get started: File:Matlab Code Following Things 2.zip. Please note that you should take any code you download from the internet (including ones from this class) with a grain of salt--you will want to verify your code is working the way it should and that any bugs are fixed. One way to help test your code is to use it on simulated data, where you know what the output should be.
- To verify that your system is sufficiently stable for accurate particle tracking, monitor a dry specimen containing 0.84μm fluorescent beads.
- Bring a slide with fixed beads into focus. Choose a field of view in which you can see at least 3 beads with the 40× objective. Limit the field of view to only those beads by choosing a region of interest (ROI) in UsefulImageAcquisition tool.
- Track the beads for 3 minutes (at least 20 fps) in a Matlab video and save the centroids with a frame rate of your choice and make a note of it.
- Use the Matlab function
track(be sure to limit the algorithm to a small region of interest around the beads, otherwise Matlab will struggle!) to separate the centroids into individual trajectories, $ \vec r_n(t) $, where $ t = nT $ and $ T $ is the inverse of the frame rate you set above.
- Use your data to calculate mean squared displacements (MSDs) of sum and difference trajectories. You may have already completed this for Problem Set 2.
- Make any necessary adjustments to your microscope and repeat this particle-tracking procedure to attain sufficient stability:
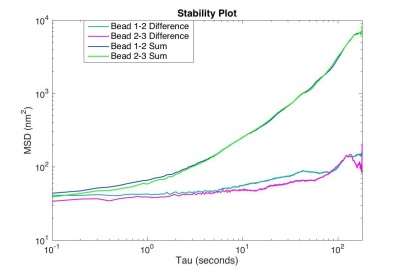
The MSD from the difference trajectory should start out less than 100 nm2 at t = 1 s and still be less than 1000 nm2 for t = 180 s.
Particle Tracking
In this part of the lab, you will follow microscopic objects throughout a series of movie frames: small, fluorescent microspheres first diffusing in purely viscous solutions of glycerol-water, and next moving in fibroblast cells after endocytosis. Calculating the mean squared displacement of their motion as a function of time interval will allow you to characterize their physical environment and behavior, first in terms of diffusivity and viscosity coefficients of the glycerol-water mixtures, next recognizing other material or transport properties in fibroblast cells.
Contextual background
Brownian motion
This section was adapted from http://labs.physics.berkeley.edu/mediawiki/index.php/Brownian_Motion_in_Cells.
If you have ever looked at an aqueous sample through a microscope, you have probably noticed that every small particle you see wiggles about continuously. Robert Brown, a British botanist, was not the first person to observe these motions, but perhaps the first person to recognize the significance of this observation. Experiments quickly established the basic features of these movements. Among other things, the magnitude of the fluctuations depended on the size of the particle, and there was no difference between "live" objects, such as plant pollen, and things such as rock dust. Apparently, finely crushed pieces of an Egyptian mummy also displayed these fluctuations.
Brown noted: [The movements] arose neither from currents in the fluid, nor from its gradual evaporation, but belonged to the particle itself.
This effect may have remained a curiosity had it not been for A. Einstein and M. Smoluchowski. They realized that these particle movements made perfect sense in the context of the then developing kinetic theory of fluids. If matter is composed of atoms that collide frequently with other atoms, they reasoned, then even relatively large objects such as pollen grains would exhibit random movements. This last sentence contains the ingredients for several Nobel prizes!
Indeed, Einstein's interpretation of Brownian motion as the outcome of continuous bombardment by atoms immediately suggested a direct test of the atomic theory of matter. Perrin received the 1926 Nobel Prize for validating Einstein's predictions, thus confirming the atomic theory of matter.
Since then, the field has exploded, and a thorough understanding of Brownian motion is essential for everything from polymer physics to biophysics, aerodynamics, and statistical mechanics. One of the aims of this lab is to directly reproduce the experiments of J. Perrin that lead to his Nobel Prize. A translation of the key work is included in the reprints folder. Have a look – he used latex spheres, and we will use polystyrene spheres, but otherwise the experiments will be identical. In addition to reproducing Perrin's results, you will probe further by looking at the effect of varying solvent molecule size.
Diffusion coefficient of microspheres in suspension
According to theory,[1][2][3][4] the mean squared displacement of a suspended particle is proportional to the time interval as: $ \left \langle {\left | \vec r(t+\tau)-\vec r(t) \right \vert}^2 \right \rangle=2Dd\tau $, where r(t) = position, d = number of dimensions, D = diffusion coefficient, and $ \tau $= time interval.
Estimating the diffusion coefficient by tracking suspended microspheres
1. Track some 0.84μm Nile Red Spherotech polystyrene beads in water-glycerin mixtures (Samples A, B and C contain 0%, 30% and 50% glycerin, respectively).
- Notes: Fluorescent microspheres have been mixed for you by the instructors into water-glycerin solutions A, B, C, and D. (a) Vortex the stock Falcon tube, and then (b) transfer the bead suspension into its imaging chamber (consisting of a microscope slide, double-sided tape delimiting a 2-mm channel, and a 22x40mm No. 1.5 coverslip, and sealed at both ends nail polish).
- Tip 1: Do not choose to monitor particles that remain stably in focus: these are likely to be 'sitting on the coverslip' and their motion will not be representative of diffusion in the viscous water-glycerol fluid.
- Tip 2: Limit the ROI to a region with only 3 or 4 particles. Long movies with the whole field of view is a sure way to make MATLAB complain.
2. Estimate the diffusion coefficient of these samples: MSD = $ \left \langle {\left | \vec r(t+\tau)-\vec r(t) \right \vert}^2 \right \rangle=2Dd\tau $, where r(t) = position, d = number of dimensions, D = diffusion coefficient, and $ \tau $= time interval. Use Sample A to verify that your algorithm correctly calculates the viscosity of water at the lab temperature (check the temperature on the clock on the wall or by other means).
- Consider how many particles you should track and for how long. What is the uncertainty in your estimate?
- From the viscosity calculation, estimate the glycerin/water weight ratio. (This chart is a useful reference. If that link doesn't work try this one.)
- See: this page for more discussion of Brownian motion and a Matlab simulation.
Live cell particle tracking of endocytosed beads
We can also use particle tracking to probe cell samples. 0.84 μm diameter red fluorescent microspheres were mixed with the growth medium and added to the plated cells for a period of 12 to 24 hours for bead endocytosis.
You will be given two plates of cells for these experiments. The cells must be imaged while they are alive, so the cells must be used the day you are given them:
- Dish 1 will be used to monitor particles in untreated cells, while
- Dish 2 will be reserved to track microspheres after adding CytoD.
- Pre-warm your DMEM++ and CytoD to 37°C
- Carefully pipet out the medium from Dish 1. Gently rinse with 1mL of medium 2X to remove beads that were not endocytosed. Then, place 2 mL of fresh medium in dish.
- Choose cells in Dish 1 with at least 2 but preferably 3 or 4 particles embedded in them and capture movies of the samples. Make sure to do this quickly, as the cells become unhealthy without the temperature and carbon dioxide regulation.
- By adjusting the LED current and exposure time, you should be able to use both bright field and fluorescence illumination simultaneously to find cells containing enough beads. Once you find a good-looking cell, turn off the LED and readjust the exposure time appropriately.
- Take as many movies as you can with about 2-5 particles in the field of view in each movie.
- Next, carefully pipet out the medium in Dish 2. Gently rinse with 1mL of medium 2X to remove beads that were not endocytosed.
- Treat the cells in Dish 2 with the cytoskeleton-modifying CytoD: Pipet out remaining medium, add 1 mL pre-warmed CytoD solution at 10 μM (pre-mixed for you) to the dish, and incubate for 20 minutes at 37°C. It's a good idea to check on your cells after 15 minutes: sometimes they are in bad shape at that point but sometimes they still look very healthy. Wash 2X with 2 mL of pre-warmed DMEM++, leaving 2 mL in the dish when imaging.
- Perform and repeat the particle tracking measurements again in Dish 2 as quickly as you are able. It would be good to image the beads in only one cell at a time, since different cells may have different degrees of cytoskeletal disruption. Take as many videos as you can before the cells become sad. The cells' physiology has now been significantly disrupted by the toxin CytoD, and they will die within a couple of hours.
Report
Find and follow all guidelines on the Microscopy report outline wiki page.
- Resolution
- Procedure
- Document the samples you used and how you captured images (camera settings, software used, etc…)
- Data
- Include an image of the PSF sample indicating which beads were used for resolution measurement..
- Include the intensity histogram of your image (created by imhist)
- Analysis and Results
- Report the resolution you measured. Make sure to include N and a measure of uncertainty.
- Show sample Gaussian fits.
- Explain the Matlab algorithm used for data analysis.
- Discussion
- Compare the measured value to the theoretical value.
- Include a thorough discussion of error sources. Do not comment on insignificant sources of error. To determine which error sources are significant, and which are not, you must think carefully about the uncertainty related to each error source and estimate its magnitude and sign. Include these estimates in your report along with your estimate of the combined, total uncertainty. It may be helpful to list out the error sources in a table, including a category for the error source, type of error (random, systematic, fundamental, technical, etc.), the magnitude of the error, and a description and way to minimize each one.
- Procedure
- Stability
- Procedure
- Document the samples you used and how you captured images (camera settings, frame rate, total number of frames, exposure, software used, etc…)
- Data
- Show an example frame from the stability movie.
- Include the MSD vs. tau plot from your stability video, containing at least 2 sum trajectories and 2 difference trajectories on a log-log scale
- Discussion
- What are the benefits and drawbacks of differential tracking?
- Include a thorough discussion of error sources.
- Procedure
- Viscosity
- Procedure
- Document the samples you prepared and used and how you captured images (camera settings including frame acquisition rate, number of frames, number of particles in the region of interest, choice of sample plane, etc)
- Data
- Include a snapshot of the 0.84 μm fluorescent beads monitored.
- Plot two or more example bead trajectories for each of the glycerin samples. (Hint: If you subtract the initial position from each trajectory, then you can plot multiple trajectories on a single set of axes.)
- Analysis and Results
- Plot the average MSD vs τ results for all glycerin samples (A, B, C, and D); use log-log axes. Use the minimum number of axes that can convey your results clearly.
- Include a table of the diffusion coefficient, viscosity and glycerin/water ratio for each of the samples (A, B, C, and D).
- Provide a bullet point outline of all calculations and data processing steps.
- Discussion
- How do your viscosity calculations compare to your expectations? (This chart is a useful reference.)
- Include a thorough discussion of error sources and the approaches to minimize them. It may be helpful to list out the error sources in a table, including a category for the error source, type of error (random, systematic, fundamental, technical, etc.), the magnitude of the error, and a description and way to minimize each one.
- Procedure
- Particle Tracking in Cells
- Procedure
- Document the samples you prepared and used and how you captured images (camera settings including frame acquisition rate, number of frames, number of particles in the region of interest, choice of sample plane, etc)
- Data
- Include a snapshot of the 0.84 μm fluorescent beads monitored.
- Plot two or more example bead trajectories for each of the samples. (Hint: If you subtract the initial position from each trajectory, then you can plot multiple trajectories on a single set of axes.)
- Analysis and Results
- Combine your data with others from the class to increase your sample size.
- Plot the average MSD (from the difference trajectories) for untreated and Cyto D treated cells on a single set of log-log axes.
- Discussion
- What kind of motion do you see described by your MSD vs τ results?
- What differences do you see between the untreated and Cyto D treated MSD curves?
- Please suggest an interpretation of the behavior of your cells based on your data.
- Include a discussion of your error sources.
- Procedure
Optical microscopy lab
Code examples and simulations
- Converting Gaussian fit to Rayleigh resolution
- MATLAB: Estimating resolution from a PSF slide image
- Matlab: Scalebars
- Calculating MSD and Diffusion Coefficients
Background reading
- Geometrical optics and ray tracing
- Physical optics and resolution
- Optical aberrations
- Aperture and field stops
- Optical detectors, noise, and the limit of detection
- Manta G032 camera measurements
- Understanding log plots
References
- ↑ A. Einstein, On the Motion of Small Particles Suspended in Liquids at Rest Required by the Molecular-Kinetic Theory of Heat, Annalen der Physik (1905).
- ↑ E. Frey and K. Kroy, Brownian motion: a paradigm of soft matter and biological physics, Ann. Phys. (2005). Published on the 100th anniversary of Einstein’s paper, this reference chronicles the history of Brownian motion from 1905 to the present.
- ↑ R. Newburgh, Einstein, Perrin, and the reality of atoms: 1905 revisited, Am. J. Phys. (2006). A modern replication of Perrin's experiment. Has a good, concise appendix with both the Einstein and Langevin derivations.
- ↑ M. Haw, Colloidal suspensions, Brownian motion, molecular reality: a short history, J. Phys. Condens. Matter (2002).