Optical Microscopy Part 1: Brightfield Microscopy
These I mention, that I may excite the World to enquire a little farther into the improvement of Sciences, and not think that either they or their predecessors have attained the utmost perfections of any one part of knowledge, and to throw off that lazy and pernicious principle, of being contented to know as much as their Fathers, Grandfathers, or great Grandfathers ever did, and to think they know enough, because they know somewhat more than the generality of the World besides:…Let us see what the improvement of Instruments can produce.
—Animadversions on the Machina Coelestis of Johannes Hevelius, 1674
Don't you just buy a [expletive deleted] microscope?
—Anonymous 20.309 student, Fall 2007
Overview
In the first week of the microscopy lab, you will construct a brightfield microscope.
Background materials and references
The following online materials provide useful background for this part of the microscopy lab.
- Geometrical optics and ray tracing
- Physical optics and resolution
- Lectures 1 through 9 of the 20.309 class
- From Nikon MicroscopyU
- Conjugate planes in optical microscopy (includes transmitted and reflected (epi) illumination)
- Snell's law
- Resolution
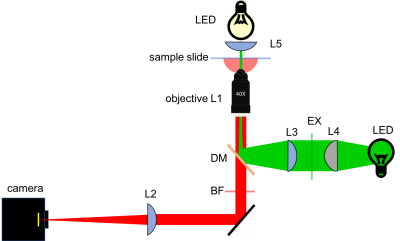
Microscope block diagram
Before you begin building, you will draw out your microscope design. To get you started, here is an example block diagram of a 20.309 microscope. Note that for Part 1, you will be building the brightfield path. You should leave space for the fluorescence path, which you will complete in Part 2.
Optical components
Below is a brief introduction to a few of the different components comprising your microscope. Various systems for optical construction are available based on rails, posts, cages, tubes, and all manner of little, metallic bits. The 20.309 microscope is constructed chiefly from cage and lens tube components made by a company called ThorLabs. The structure should be rigid, and the components sufficiently tightened so that your optics remain aligned even after moving your microscope. Understanding how all of the components work together can be daunting. Ask about any components that perplex you.
Lenses
Plano-convex spherical lenses are available with focal lengths of 25, 50, 75, 100, 125, 150, 175, and 200 mm. Plano-concave lenses with focal lengths of -30 and -50 are also available. It is best to mount most optics in short (e.g. 0.5") lens tubes. It is acceptable to mount a lens between the end of a tube and a tube ring or between two tube rings. In most cases, the convex side of the lens faces toward the collimated beam; the planar side goes toward the convergent rays.
- Tip: Verify all optics before you use them by determining the focal length with a ruler. You can use the lens measuring station. Alternatively, you can use the ceiling fluorescent lamps as a light source and measure the exact distance between the lens(es) assessed and the lamp's image. Can you imagine a simple rig to evaluate negative focal lengths (of plano-concave lenses for instance)?
- Tip: As you install lenses into your microscope, put a piece of tape on the lens tube showing focal length and orientation. This will help you both during construction and put-away. Save the lens storage boxes and return components to the correct boxes when you are done.
- Handle lenses only by the edges. If a lens is dirty, first remove grit with a blast of clean air or CO2. Clean the lens by wiping with a folded piece of lens paper wetted with a drop of methanol. (Do not touch the part of the tissue you use for cleaning with your fingers.) In some cases, it may be helpful to hold the folded lens tissue in a hemostat. Ask an instructor if you need help.
Objective lenses
Please see the Nikon Introduction to Microscope Objectives at their excellent MicroscopyU website.
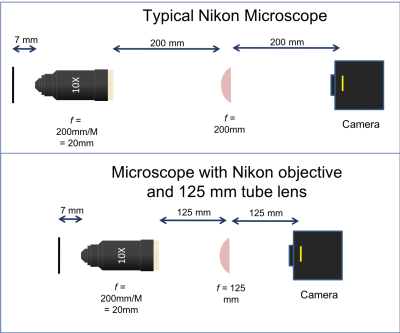
There are three objective lenses available in the lab: a 10×, a 40×, and a 100×. All of these are designed to use a 200 mm tube lens to form an image on the camera. An adapter ring converts the objective mounting threads to the SM1 threads used by the lens tube system.
- Working distance (WD) is the distance between the front objective lens surface and the cover slip, and so it is also approximately the distance to the front focal plane. In order to focus an image at the back focal plane of the tube lens, i.e., on the CCD array, the sample plane must coincide with the front focal plane in a 4f microscope arrangement. The stage is added to hold the sample in this plane.
- The 100× objective is designed to be used with immersion oil. When using the 100× objective, place a drop of oil directly on the tip on the objective. Bring the drop in contact with the slide cover glass. After use, clean off the remaining oil by wicking it away with lens paper or a Kim-wipe. Do not put samples away dirty.
- Note that the back focal plane (BFP) of the objective does not necessarily coincide with the rear of the objective housing. In fact, for the Nikon 40x objective the BFP is close to the blue ring. You will find its actual location when aligning the laser path in Part 2. The 200 mm distance labeled between the back of the objective housing and the tube lens is a recommendation from Nikon to enable optimal imaging. For details on the importance and origin of this distance please ask an instructor.
Sample stage
A precision Newport X/Y/Z stage[1] with a sample holder mounted on a post, or a Thorlabs Max312D stage, also with a sample holder, is available at each lab station. The Newport stage setup is top-heavy. Avoid accidents by ensuring that the post base is always attached to an optical breadboard or table. Leave the stage at the lab station when you are done with it. For the Thorlabs stages, it is still a good idea to bolt them down so that your area of interest (AOI) stays in your microscope field of view (FOV).
All stage axes have limited adjustment range, especially the Thorlabs stages. To deal with this, it is best to leave the stage base bolts and sample holder bolts loose and move the sample holder in x, y and z to roughly find your AOI. Once you are on or near your AOI, tighten the bolts and use the micrometers to center your image. One trick here is to get the z clamped first, then deal with x and y.
CCD camera
The microscope you will build does not have an eyepiece for direct visual observation. Instead, images will be captured with a CCD camera[2]. Its monochrome (black and white) sensor contains a grid of 656×492 square pixels that measure 7.4 μm on a side. An adapter ring converts the C-mount thread on the camera to SM1.
Microscope construction
Design
Sketch out the design for your microscope on paper. For this part of the lab, you can just draw the bright field illumination path. Include all the optical elements (lenses, mirrors, microscope objectives, and camera). Label all distances, lens specifications, and orientations. You will include this diagram (or a cleaned-up version) in your lab report.
- Some elements must be positioned precise distances apart; other distances are not critical. Use ray-tracing to determine when this is the case. Which distances in your bright-field microscope will be critical? Which will be forgiving or unessential? Which will change with each objective lens (10×, 40× and 100×)?
- Which sections of the light path can be open (strut-based structure, cage rods)? Which would better enclosed (Thorlabs lens tubes)?
- In what way will the illumination LED color affect your design? your results?
- Which lens will you use between the LED and the sample for bright field transmitted light imaging?
Practice
- On a 1' x 2' x 1/2" optical breadboard, build a bright-field imaging microscope, using the provided LED as a light source, CCD camera as a light detector, and the rigid mounting components and lenses at your disposal.
- You may view the example microscope for reference. However, please do not touch, alter, or remove parts from the example microscope.
- Even though you're first focusing on the bright-field imaging leg of your microscope, take into consideration some requirements pertinent to the fluorescence imaging elements you'll add to your system next week:
- Reproduce the general layout of the example microscope: it grants compactness and allows your device to be a stand-alone breadboard-transportable microscope.
- Set the distance between the top of the breadboard and the top surface of the upper LCP01 to 13.5 cm. It is important to ensure your construction is compatible with either of the two distinct stage mounting platforms available in the 20.309 lab (either Newport or Thorlabs model). If you find it inconvenient to measure this, there is a Handy Scope Height Thingama-jig floating around the lab. Ask your instructor(s). Also, note that the stages are very expensive; always lift them from the bottom.
- Align the vertical Thorlabs P14 (1.5" diameter mounting post) with a breadboard hole that is 11 positions from a short side and 5 positions from a long side. This allows enough free space on the breadboard such that either the Newport or the Thorlabs stages may be utilized. (Note that the picture is in error as the P14 is only 9 positions from a short side of the breadboard.)
- Reproduce the general layout of the example microscope: it grants compactness and allows your device to be a stand-alone breadboard-transportable microscope.
- Do insert the C6W cage cube that will later hold the dichroic mirror on while fluorescence imaging will rely. Be sure to keep the mounting struts fully recessed in the cube walls; their ends should not stick out, they would otherwise hinder maneuvers with dichroic-holding kinematic plate!
- Verify the focal length of the lenses you selected. If you find an optic in the wrong box: identify the optic and replace it in the correct box or label the box correctly. (Ask an instructor if you can't find the right box. There are many boxes near the wire spools behind you as you stand at the wet bench.)
- Check all your lenses for cleanliness before you use them. You'll save yourself some troubleshooting time and effort down the road!
- Make sure all your components are "leveled" (horizontal, not slanted).
- Use tube rings (and never an SM1T2, SM1V01, or SM1V05) to mount optics in lens tubes.
- Use adjustable mounting components in front of the CCD camera so you can optimize and fine-tune the camera positing with respect to the imaging lens L2. Beware: never use an SM1T2 coupler without a locking ring — they are very difficult to remove if they are tightened against a lens tube or tube ring. Also put a quick-connect in your design such that the camera CCD will end up 200 mm from the back focal plane of the objective. Remember that the CCD is recessed inside the opening of the camera.
- Use only three cage rails to connect the C6W cage cube and the KCB containing the last silver mirror before the CCD camera, so you can easily take in and out the barrier filter (BF) that will later aid fluorescence-mode microscopy. Always place two rails at the top so that an alignment target can be hung if needed (the benefit will become more clear in Part 2).
- The Nikon objective lenses are designed to be paired with a 200 mm tube lens.
- Assume that the objectives behave as ideal plano-convex lenses.
- Fine focusing will be achieved by adjusting the height of the sample stage.
- Tip: Throughout the optical microscopy lab, start the alignment with a 10× objective and then progress to 40× and 100×.
- A red or a blue LED illuminator can be used for bright-field transmitted light imaging. On one hand a blue LED yields a better bright-field resolution, however bright-field resolution is not usually critical in this lab. On the other hand, a red LED allows simultaneous fluorescent and bright-field imaging in Parts 2 and 3, and this can be quite useful when trying to bring a fluorescent sample into focus.
- Each group will receive their own LED. Please ask an instructor if you cannot find one.
| |
Double check your wiring before powering the LED. The LED can be damaged by excessive current. Limit the driving current to 0.5 A to protect the LED. |
Recording, displaying and saving images in MATLAB
- Run the UsefulAcquisition tool
- Launch MATLAB and in the command window type:
foo = UsefulImageAcquisition; foo.Initialize
- The Image Acquisition window (Figure 1) will open with the controls for the camera.
- The "Manta_G-032B" camera is configured to produce 12-bit, monochrome images. In this mode, the intensity of each pixel in the image will be represented by 12 binary digits, allowing a range of values from 0-4095.
- In the Image Acquisition window set "Frame Rate" to 20. This will cause the camera to take 20 complete images per second (this will only be relevant when recording movies in Part 3 of the lab).
- Click the "Start Preview" button. The live image from the camera should appear in the Preview pane.
- If this does not produce a live image, use an appropriate expletive, and ask an instructor to figure out what the heck went wrong.
- Change the "Exposure" setting to produce a good image. The value sets the exposure time for each frame in microseconds.
- Recording an image
- In the Image Acquisition window, set "Number of Frames" to 1. This setting controls how many images MATLAB will record each time you press the "Acquire" button.
- When you are happy with the image displayed by the live preview, press "Acquire". The live preview will stop.
- The image is now stored in the foo.ImageData variable, which will update each time you acquire a new image. To copy the data into a new variable, choose a descriptive name for your image like 'microruler10x' and save it to your workspace by typing the following into the MATLAB command window:
microruler10x = foo.ImageData;
- Next, in the MATLAB command window type
- This command will display relevant information about the new variable you’ve created. You should see that the image is represented as a 492x656 matrix of 16-bit integers.
- Displaying the image
- You can display images using a variety of commands in MATLAB. In the optics bootcamp we used the imagesc command which scales the image intensity (the brightest pixel is white, the darkest is black). In some cases, like when you have very dim images, this command can be misleading. It’s better to use the un-scaled imshow command for quantitative measurements.
- When the 12-bit numbers from the camera get transferred to the computer, they are converted to 16-bit numbers. 16-bit numbers can represent a range of values from 0-65535, while your 12-bit image only contains values from 0-4095. This leaves a considerable portion of the number range unoccupied. Consequently, if you type imshow( microruler10x ), you will see an image that looks almost completely black (try it!).
- One way to make this work is to rescale your image to 16 bits: imshow( 65535/4095 * microruler10x )
- An even better way to work with images in MATLAB is to convert them to double precision floating point format straightaway. Double precision floating point numbers can represent an extremely wide range of values with high precision. Convert the image to a double and rescale it using the following command:
microruler10x = double( microruler10x ) / 4095;
- This conversion has made your image into a double with a range of intensities from 0-1, with 1 being full intensity and 0 completely dark. Now try:
- Saving your image
- Save images in a .mat format so that you can easily reload them into Matlab for later use.
save microrulerImages % saves entire workspace to filename 'microrulerImages.mat' save microrulerImages image1 image2 %saves only variables image1 and image2 to filename 'microrulerImages.mat'
- To reload your data the next time you open matlab, navigate to the folder where you saved your workspace, type
- If you want to save individual images as a .PNG (a good format for use in your report or other programs), the command might look something like:
- Note that you can also use the File→Save As menu after displaying an image or figure. This is useful if you want to save additions to your images (like data cursors). However, we recommend always saving the raw data as .mat files so that you are able to re-process your images later.
whos microruler10x
whos microruler10x imshow( microruler10x )
load microrulerImages
imwrite( im2uint16( microruler10x ), 'microruler10x.png', 'png' );
Magnification measurement
Measuring the magnification of your microscope is a good way to verify that your instrument is functioning well. You should measure the magnification of any microscope you plan to use for making quantitative measurements of size. Use the measured value in your calculations, not the number printed on the objective. Consider the uncertainty in your measurement.
- Start the live preview using the UsefulImageAcquisition tool
- Ensure that the camera's field of view is approximately centered in the objective's field of view.
- The objective has a larger FOV than the camera. Use the adjustment knobs on mirror M1 to traverse the objective's FOV horizontally and vertically. The FOV is approximately circular. Find a spot near the middle.
- Start with the 10x objective and a microruler calibration slide.
- The microruler calibration slide has tick marks that are 10 um apart. Every 100 um, there is a longer tick mark.
- Make sure that the side of the microruler with the pattern on it faces the objective. Imaging through the thick glass causes distortion and many other troubles.
- Record an image of the microruler.
- Use imdistline or the data cursor to measure a known distance between rulings in your image and compute the magnification.
- When choosing a distance to measure, consider the factors that influence the uncertainty of your measurement.
- Repeat the magnification measurement for the 40x and 100x objectives.
- With the 100x objective, you may want to substitute the microruler with a Ronchi Ruling, a grating with 600 line pairs per millimeter. Why is it not wise to use the Ronchi Ruling with the 10x objective?
- Save your images in a .mat file for later use in MATLAB or as a PNG image for use in your report or other programs.
- Using your magnification measurements, calculate the FOV of the microscope for each objective.
Particle size measurement
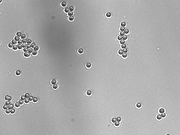
Now that you know the magnification of your instrument, use it to measure the size of some microscopic objects as imaged with the 40x objective lens only. Slides with 7.2 μm, 3.2 μm and 1 μm silica microspheres are available in the lab.
- Image 7.2 μm, 3.2 μm and 1 μm silica microspheres as described in the magnification measurement procedure (40x objective only).
- Measure and report the average size and uncertainty of the spheres in each sample. How many spheres should you measure?
Microscope storage
During the microscopy lab, approximately seven thousand optical components will be taken from stock, assembled into microscopes, and properly returned to their assigned places. Please observe the following:
- Store your microscope in one of the cubby holes in 16-336 (not in the lab). If you use one of the high shelves, get somebody to help you lift.
- Keep all of the boxes for the optics you use with your instrument to simplify putting things away.
- Take a blue bin to store loose items (such as lens boxes) in.
- Stages, CCD cameras, neutral density filters and barrier filters stay at the lab station. Do not store these with your microscope.
- Return objective lenses to the drawer when you are not using them. (Do not store them with your microscope.)
- The stages are very expensive. Always lift from the bottom.
- If you break something (or discover something pre-broken for you), do not return it to the component stock. Give all broken items to an instructor. You will not be penalized for breaking something, but not reporting may be looked upon less kindly.
Report requirements
General guidelines
- Each part of the microscopy lab requires its own submission. Only the final report will include all 3 sections. You may revise any part of your report until the final deadline. Update the apparatus section to reflect any changes you make during the course of the lab.
- On each due date, one member of your group should submit a single PDF file to Stellar in advance of the deadline. The filename should consist of the last names of all group members, CamelCased, in alphabetical order, followed by a hyphen, the name of the assignment, with a .pdf extension. Example: CrickFranklinWatson-MicroscopyPart1.pdf.
- Use the outline below to break your report into sections.
- Keep your lab report short. Use bullet points, tables, and other organizational tools when appropriate. Brevity is not an excuse for imprecise, incomplete, or unclear communication.
- Be selective in what images you include in your report. Where images are required, include one or a few that convey the character of the dataset. Size images appropriately (does it really need to take up half a page?). At the same time, ensure plots images are readable. A good trick for reducing plot size while maintaining clarity is to adjust the font size and line values so the plot remains clear even when it is small. It helps to save images and plots in an uncompressed format; import and resize them in a way that retains the full quality; and create your final PDF file with settings that do not compress plots and pictures.
- An outstanding error discussion is an essential element of a top-notch report.
- Include any computer code you developed in an appendix at the end of your report. Indicate the source of any code used that you obtained from outside sources.
- Refer to this page for more detail on how to write an excellent report, and to this page for an outline of the final microscopy report.
Part 1 report outline
- Apparatus:
- Include a block diagram of your microscope, including all optical elements and relevant distances. It is not necessary to document the details of the mechanical construction.
- Describe your design calculations and considerations.
- Why not put in a nice snapshot of your ‘scope? (optional, but certainly a cherished memory in the making)
- Magnification
- Procedure
- Document the samples you used and how you captured images (camera settings, software used, etc…)
- Data
- Include example images.
- Analysis and Results
- Report the nominal and actual magnifications and fields of view you measured for the three objectives in a table. Report the length and width of the FOV (in distance units), not its area (in distance units squared).
- Document the method you used to find magnification.
- List the error sources that contributed significantly to systematic error and uncertainty in your measurement. To the degree possible, quantify the type and magnitude of the error.
- Discussion (optional for magnification measurement)
- Explain any challenges you faced in the magnification measurement.
- Procedure
- Particle diameter measurement
- Procedure
- Document the samples you used and how you captured images (camera settings, software used, etc…)
- Data
- Include example images.
- Analysis and Results
- Report the average size of the microspheres in each sample and a measure of variation.
- Describe how you measured the microspheres.
- List the error sources that contributed significantly to systematic error and uncertainty in your measurement. To the degree possible, quantify the type and magnitude of the error.
- Discussion
- Explain any challenges you faced measuring the size of silica microspheres.
- How did your measurements differ from the manufacturer's specified values? What factors contributed to the difference?
- Procedure
Optical microscopy lab
Code examples and simulations
- Converting Gaussian fit to Rayleigh resolution
- MATLAB: Estimating resolution from a PSF slide image
- Matlab: Scalebars
- Calculating MSD and Diffusion Coefficients
Background reading
- Geometrical optics and ray tracing
- Physical optics and resolution
- Optical aberrations
- Aperture and field stops
- Optical detectors, noise, and the limit of detection
- Manta G032 camera measurements
- Understanding log plots