20.109(F22):M1D1
Contents
Introduction
In the past century, we have learned a tremendous amount by studying the behavior of mammalian cells maintained in the laboratory. Tissue culture was originally developed about 100 years ago as a method for learning about mammalian biology. The term tissue culture was coined because people were doing exactly that, extracting tissue and letting it live in a dish for a short time. Today, most tissue culture experiments are done using isolated cells rather than whole tissues. Much of what we know about cancer, heritable diseases, and the effects of the environment on human health has been derived from studies of cultured cells.
Cells that are isolated from tissue are called primary cells, because they come directly from an animal. It is very difficult to culture primary cells, largely because primary cells that are grown in culture divide only a limited number of times. This limitation on the lifespan of cultured primary cells, called the Hayflick limit, is a problem because it requires a researcher to constantly remove tissues from animals in order to complete a study. Cell isolation processes can be quite labor-intensive, and also can complicate data analysis due to inherent animal-to-animal variation. To get around the first of these problems, researchers use cells that are immortal, which means they can divide indefinitely, though some inherent cell-to-cell variation still exists in such cells.
One familiar type of immortalized cell is the cancer cell. Tumor cells continuously divide, allowing cancer to invade tissues and proliferate. Cancer cells behave the same way in culture, and under the right conditions, cells can be taken from a tumor and divide indefinitely in culture. Another type of immortalized cell is the embryonic stem cell. Embryonic stem cells are derived from an early stage embryo, and these cells are completely undifferentiated and pluripotent, which means that under the right conditions, they can become any mammalian cell type.
The art of tissue culture lies in the ability to create conditions that are similar to what a cell would experience in an animal, namely 37 °C and neutral pH. Blood nourishes the cells in an animal, and blood components are used to feed cells in culture. Serum, the cell-free (and clotting-factor free) component of blood, contains many of the factors necessary to support the growth of cells outside the animal. Consequently, serum is frequently added to tissue culture medium, although serum-free media exist and support some types of cultured cells. Furthermore, cultured mammalian cells must grow in a germ-free environment and researchers using tissue culture must be skilled in sterile technique.
Protocols
Part 1: Complete Orientation quiz
Complete the orientation quiz with your partner. Though you are working with your partner, each student should record all answers independently. If you disagree with your partner on an answer, you should write what you think is the correct answer on your quiz.
Good luck!
Part 2: Learn cell culture best practices
One major objective for this experimental module is for you to learn best practices for cell culture using correct sterile techniques. Pay close attention to the demonstration provided by the Instructor!
Review the following resource before you complete the tasks detailed in this exercise: Guidelines for working in the tissue culture room
Preparing tissue culture hood
- The tissue culture hood is partly set up for you. Finish preparing your hood according to the demonstration, first bringing in any remaining supplies you will need, then obtaining the pre-warmed reagents from the water bath, and finally retrieving your cells from the 37 °C incubator.
- Be sure to spray everything (except cells) with 70% ethanol and wipe dry before moving items into the tissue culture hood!
- One of the greatest sources for tissue culture contamination is moving materials in and out of the hood because this disturbs the air flow that maintains a sterile environment inside the hood. Think about what you will need during your experiment to avoid moving your arms in and out of the hood while you are handling your cells.
Collecting cells
- Obtain one ~48 h cultures of MEF cells in T25 flask from the 37 °C incubator.
- Examine your cell cultures after you remove the flask from the incubator.
- Look first at the color and clarity of the media. Fresh media is reddish-orange in color and if the media in your flask is yellow or cloudy, it could mean that the cells are overgrown, contaminated, or starved for CO2.
- Next, look at the cells using the inverted microscope. Note their shape, arrangement, and how densely the cells cover the surface of the flask.
- After you look at your cells, take the flask to your tissue culture hood.
- Wash the cells by adding 5 mL PBS using a 5 mL pipet. Slightly tip the flask back and forth to rinse the cells then aspirate the PBS with a fresh Pasteur pipet.
- To dislodge the cells from the flask, you will add trypsin which is a proteolytic enzyme.
- With a 2 mL pipet, add 1 mL of trypsin to the flask.
- Be careful with 2 mL pipets as they fill quickly and liquid will go all the way up your pipet into the pipet-aid. If this happens, please alert the teaching faculty rather than returning the pipet-aid to the rack.
- Tip the flask in each direction to distribute the trypsin evenly then incubate the cells at 37 °C for 2 minutes.
- This is a great time to clear out your trash and read ahead!
- Retrieve your flask from the incubator and firmly tap the bottom to dislodge the cells.
- Check your cells using the microscope to ensure they are dislodged. They should appear round and move freely.
- If your cells are not detached from the flask, incubate at 37 °C for an additional minute.
- When your cells are dislodged, move your flask back into the tissue culture hood and add 5 mL of media to the cells then pipet the liquid up and down (“triturate”) to break up cells that are clumped together and suspend them in the liquid.
- Note: do not take up or release all the liquid, in order to avoid bubbles.
- Transfer the suspended cells into a labeled 15 mL conical tube.
- Transfer 90 μL of your cell suspension from the 15 mL conical tube into a labeled eppendorf tube.
Counting cells
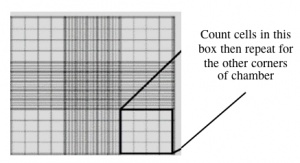
During your work in tissue culture, you will use a hemocytometer to count mammalian cells. More importantly, you will use the cell count information to determine the density of your cultures. A hemocytometer is a modified glass microscope slide that has a chamber engraved with a grid. Stained mammalian cells are loaded into the chamber, which is manufactured such that the area within the gridlines is known and the volume of the chamber is known. These features enable researchers to count the number of cells within a specific volume of liquid.
Using a hemocytometer, you can determine the density (cells per mL) of cell culture.
- Carry the tube with your 90 μL cell suspension aliquot to the center microscope bench and add 10 μL of trypan blue cell stain. Mix by pipetting up and down.
- Carefully pipet 10 μL of the stained cells between the hemocytometer and (weighted) glass cover slip.
- Count the cells that fall within the four corner squares (with a 4 x 4 etched grid pattern), average (i.e. divide by 4), and then multiply by 10,000 to determine the number of cells/mL.
In your laboratory notebook, complete the following:
- Use the information provided to calculate the density of the cell culture.
- Calculate the total number of cells that were collected.
- Hint: use the density calculated and the total volume for this calculation.
- Calculate the volume of cells required to prepare a 15 mL cell suspension at a final density of 25000 cells/mL.
- You will prepare and use this cell suspension in Part 3.
Part 3: Seed cells for gamma-H2AX assay
- Obtain one 12-well plate and coverslips from the center laboratory bench.
- Clearly label the plate with the date and your team name. Include the name of the cells seeded!
- Carefully place one coverslip into each of the wells of the plate.
- Prepare your cell suspension using the calculations from Part 2.
- Transfer the appropriate volume of cells to a 50 mL conical tube.
- Add fresh media such that the final volume is 15 mL.
- Add 1 mL of the cell suspension into each of the wells in the 12-well plate.
- Cells settle quickly in conical tubes so it is important you mix before adding cells to the wells!
- Give any extra cell suspension to the Instructor.
- Carefully move your plate to the 37°C incubator
- Clean out the tissue culture hood:
- Aspirate any remaining liquids.
- Dispose of all vessels that held cells in the biohazard waste box and be sure that all sharps are in the sharps jar.
- Remove any equipment or supplies that you transferred into the hood and return to the appropriate location.
- Please leave the equipment that was already there.
- Spray the TC hood surface with 70% ethanol and wipe with paper towels.
- Be sure the paper towels are disposed of in the biohazard waste box!
- Empty the benchtop biohazard bucket into the biohazard waste box.
Part 4: Research MEF cells
For the experiments in this module, you will use mouse embryonic fibroblast (MEF) cells. The specific MEF cells that will be used are primary cells that were collected by researchers in the Engelward Laboratory.
To familiarize yourself with this type of cell, you and your laboratory partner will research the questions below.
In your laboratory notebook, complete the following:
- What is the difference between primary cells and a cell line?
- What is one advantage and one disadvantage of using primary cells in experiments? For using a cell line?
- Why is it important to track the number of passages when working with primary cells?
Reagents list
- Mouse embryonic fibroblast (MEF) cells (a gift from the Engelward Laboratory)
- Dulbecco's Modified Eagle Medium (DMEM) (from Gibco), supplemented with:
- 20% fetal bovine serum (FBS) (from Atlanta Biologicals)
- 100 U/mL of antibiotic solution, containing penicillin and streptomycin (from Gibco)
- trypan blue (from VWR)
- incubator maintains 37°C, 5% CO2 and 95% relative humidity
Next day: Prepare and treat cells for γH2AX experiment