20.109(F20):M3D5
Contents
Introduction
Congratulations on reaching your final (virtual) laboratory day in 20.109! To complete your experience and training, the goal for today to synthesize the data that you collected and analyzed throughout this module and refine the approach such that an improved hypothesis can be tested. By pulling all of this information together you will, hopefully, be able to further improve ethanol yield in E. coli MG1655 using the CRISPRi system. As discussed at the start of Module 3, the basis of scientific research is an iterative process that consists of four stages: designing experiments, collecting data, analyzing results, and refining the approach. This rigorous cycle is how we ensure our results are accurate and reproducible!
When considering results that will be used to refine a research approach, it is important to recognize that not all data are created equal. For several reasons, including technical error and reagent failure, it often happens that an experiment does not work as expected. By including controls, researchers are able to identify these issues and rectify them in follow-up experiments. In addition to using controls that validate the results, researchers use replicates and repeat experiments to ensure the data are robust. All of these internal checks allow researchers to be confident about the results they report.
Today you will critically think about the data that you analyzed in this module and design a 'better' sgRNA_target molecule given what you learned in your research. Though considering which of the current sgRNA_target sequences yielded the highest level of ethanol is important in your goal for today, it is just as important to decide which experiments produced results that are valid when designing your optimized targeting sequence.
Though you will not have the opportunity to use your optimized sgRNA_target molecule this semester, you are more than welcome to complete the ethanol yield experiment with your sgRNA_target as soon as you are invited back to campus! We will be more than happy to host you in the laboratory for some actual benchwork!!
Protocols
Part 1: Determine which target is most effective for increasing ethanol yield
Your first task for today is to review the data analysis you completed on M3D3 and decide which datasets you will consider when designing your optimized sgRNA_target. You should base this decision on the following: 1. the reproducibility of the replicates and 2. the validity of the controls.
Next, you will decide on the approach you will use to optimize ethanol yield! After choosing which gene you think is the best target for increasing ethanol yield, consider how to improve upon the sgRNA_target sequences that were created / tested by previous 109ers. Recall the research you completed on M3D4 when discussing which sequence in the genome to target.
In your laboratory notebook, complete the following with your partner:
- Decide on a target.
- Which gene you will target (ldhA or pta-ack)?
- Will target the promoter or the coding region of the gene you selected.
- Provide the rational for your design choice.
- Why do you think the target gene you selected is better than the other option?
- Why do you think targeting the promoter / coding region is best?
Part 2: Design optimized sgRNA_target for improved ethanol yield
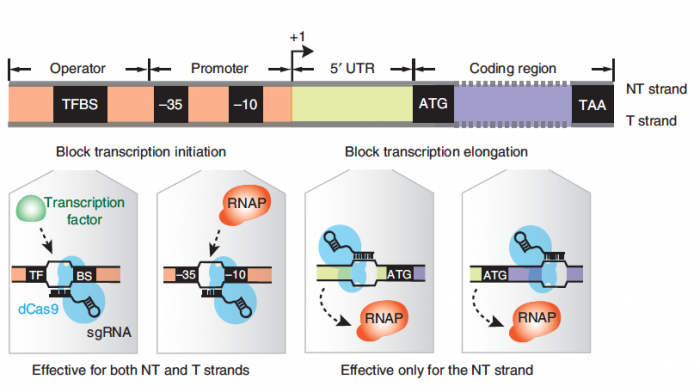
Now that have a strategy for optimizing ethanol yield using the CRIPSRi system, you need to design a sgRNA molcule using the DNA sequence of the target gene. As a reminder, the sgRNA recruits dCas9 to a specific location in the genome where it will bind and block transcription. The CRISPRi system can block transcription at either the initiation or elongation steps of transcription as illustrated in the figure below. If the dCas9 protein is bound to the promoter sequence at either a transcription factor binding site (TFBS) or the -10 / -35 boxes it will block transcription factors and / or RNA polymerase from binding to the promoter of the gene, thereby impeding transcription initiation. If the dCas9 protein is bound within the gene it will impede RNA polymerase elongation of the transcript, or if dCas9 is bound within the gene at a TFBS transcription could be inhibited due the role of the TF in gene expression. Based on your analysis of the previously collected ethanol yield data and your assessment of the genetic features present in the promoter / gene, which approach is best?
As you consider how to best optimize a sgRNA_target sequence, keep in mind that you have freedom to design an sgRNA_target sequence that you think will best increase ethanol yield. You can modify one of the existing sgRNA_target sequences from the data you analyzed in the module, or you can design an sgRNA_target from scratch using the information you gathered in your analysis of the data. When you decide on which region of the target gene you will use, design your optimized sgRNA_target sequence according to the steps below:
- Use the KEGG Database to obtain the DNA sequence of the target gene in the E. coli K-12 MG1655 strain.
- Enter the name of your target gene in the Search genes box and click Go.
- Double click on the linked gene name.
- Determine the position of your gene within the genome by clicking the Genome map button within the Position row.
- Is your target gene at the start of an operon or in the middle?
- Does the position of your gene within the operon affect the sgRNA targeting strategy you discussed with your partner? If you are unsure, consult with the Instructors.
- The amino acid (AA) sequence and nucleotide sequence (NT) of your gene are provided at the bottom of the page.
- Generate a new DNA file in SnapGene that contains the NT sequence of your gene.
- If you will design an sgRNA_target that binds the promoter, enter 50 in the +upstream box to get the 50 basepair sequence immediately preceding the start codon.
- Use the DNA sequence to select a 20-25 basepair region that will serve as the sgRNA_target binding site. Please make sure your sequence does not exceed 25 bp.
- If you target the template DNA strand, the sgRNA_target sequence will be the same as the transcribed sequence.
- If you target the nontemplate strand, the sgRNA_target sequence will be the reverse-complement of the transcribed sequence.
- To avoid off-target effects, check the specificity of your sgRNA_target sequence using the Basic Local Alignment Search Tool (BLAST) available from NCBI.
- Click on the Nucleotide BLAST icon.
- In the Enter Query Sequence section, paste your sgRNA_target sequence into the Enter accession number(s), gi(s), or FASTA sequence(s) box.
- In the Choose Search Set section, type 'MG1655' in the Organism box and select an appropriate option from the drop down list.
- Click the BLAST button at the bottom of the screen.
- Ideally, your sgRNA_target sequence will only have 100% identity to the target sequence.
- If your sgRNA_target sequence binds to multiple locations (with a 14+ nucleotide region within the gRNA having 100% identity to an off-target) within the genome, consider redesigning your sgRNA_target.
In your laboratory notebook, complete the following:
- Record your sgRNA_target sequence.
- Be sure you write your gRNA sequence 5' → 3'.
- Recall that additional sequence information must be added to your sgRNA_target for the CRISPRi system. A 'handle' is required for dCas9 binding to the sgRNA. The tag sequence 5' - GTTTTAGAGCTAGAAATAGCAAGTTAAAATAAGGC - 3' should be included at the 3' end of your sgRNA_target. Please double check that in total, the full sequence is 60 bp or fewer.
- Provide the rational for your design choice.
- Why did you choose to target the specific basepair sequence in the promoter / coding region?