20.109(F20):M2D2
Contents
Introduction
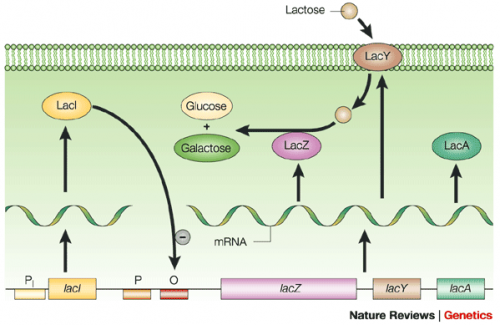
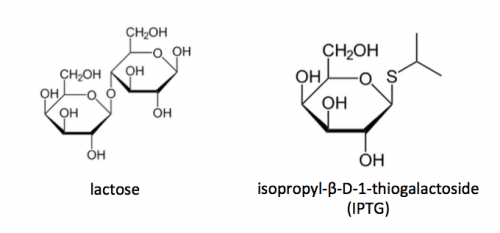
To induce production of PF3D7_1351100 protein from the expression plasmid that was 'cloned' in the previous laboratory session, a lactose-analogue isopropyl β-D-1-thiogalactopyranoside (IPTG) was used to induce expression in Nico(DE3) E. coli bacterial cells. The use of IPTG to induce protein expression is based on the native lac operon used for lactose metabolism in bacterial cells.
The lac operon is composed of four genes: lacI, lacZ, lacY, and lacA. When lactose is absent, LacI (the protein encoded by lacI) binds to the operator sequence (O) upstream of lacZYA. In the presence of lactose, LacI and lactose form a complex which relieves repression of lacZYA transcription. LacZ is a β-galactosidase that cleaves lactose resulting in glucose and galactose. LacY, a β-galactoside permease, facilitates the transport of lactose across the cell membrane, and LacA, a β-galactoside transacetylase, transfers an acetyl group from acetyl-CoA to β-galactosides.The native lac operon is a powerful tool in engineering protein expression systems because it enables researchers to control gene expression using inducer molecules. The lacZYA genes are only expressed when lactose is present. If a gene of interest is cloned downstream of the operator sequence, the expression of this gene can be controlled by LacI repression and lactose derepression. To further control the system for protein expression, IPTG is used as a lactose-analog as it is not metabolized by the cells.
Today you will isolate the expressed PF3D7_1351100 protein from the bacterial cells. Remember that the PF3D7_1351100 gene sequence was synthesized using a gBlock and 6xHis-tag was added to the DNA sequence. The resultant protein is therefore His-tagged. Histidine has several interesting properties, notably its near-neutral pKa, and His-rich peptides are promiscuous binders, particularly to metals. (For example, histidine side chains help coordinate iron molecules in hemoglobin.)
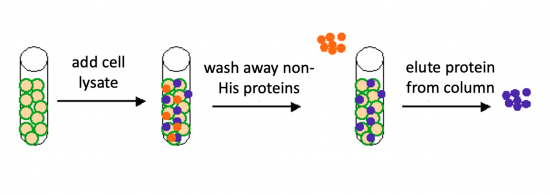
To purify the PF3D7_1351100 proteins present in the bacterial cell, you will use a nickel-agarose resin. The 6xHis-tag on the PF3D7_1351100 protein will bind to the nickel-coated resin, while the other cellular protein will pass through the resin. Remember, the Nico(DE3) cells are not only producing the PF3D7_1351100 protein, but also the proteins needed for cellular function and survival. Imidazole is a compound that is also able to bind to nickel and washing the resin with a low concentration solution promotes competition for binding between the imidazole and bound proteins for the nickel-coated resin. Proteins that are non-specifically bound will have a lower affinity for the nickel than imidazole and be washed from the column, whereas the 6xHis-tagged PF3D7_1351100 will remain adhered to the nickel-agarose resin. To elute the PF3D7_1351100 protein from the nickel-coated resin, a high concentration of imidazole is used to out-compete the 6xHis-tag for binding.
Protocols
Part 1: Induce expression of PF3D7_1351100
To ensure the steps included below are clear, please watch the video tutorial linked here: [Bacterial Induction]. The steps are detailed below so you can follow along!
- Inoculated 5 mL of TB media containing 50 μg/mL kanamycin with a colony of Nico(DE3) cells transformed with pET-28b(+)_PF3D7_1351100.
- Incubated the culture overnight at 37 °C with shaking at 220 rpm.
- Dilute the overnight culture 1:100 in 100 mL of fresh TB media containing 50 μg/mL kanamycin.
- Incubate at 37 °C until the OD600 = 0.6 with shaking at 220 rpm.
- To induce PF3D7_1351100 protein expression, add IPTG to a final concentration of 1 mM.
- Incubate at overnight at room temperature with shaking at 100 rpm.
- To harvest the cells, centrifuge the culture at 4000 g for 15 min at 4 °C.
- Cell pellets were flash frozen in liquid nitrogen, then stored at -80 °C until used for purification.
In your laboratory notebook, complete the following:
- Calculate the volume of kanamycin stock that was added to the TB broth in Step #1. In Step #3.
- Concentration of kanamycin stock = 50 mg/mL.
- Calculate the volume of IPTG stock that was added to the TB broth in Step #5.
- Concentration of IPTG stock = 100 mM.
Part 2: Purify PF3D7_1351100 protein
To ensure the steps required for purifying the PF3D7_1351100 protein are clear, the Instructor will provide a live demonstration of this process. You should include notes regarding the procedure in your laboratory notebook!
Lyse Nico(DE3) cells expressing pET-28b(+)_PF3D7_1351100
- Retrieve the Nico(DE3) pET-28b(+)_PF3D7_1351100 cell pellet from the -80 °C freezer and leave it on your bench to thaw.
- Add the cell lysis buffer and components to each cell pellet.
- B-Per bacterial extraction reagent at 4 mL / g of cell pellet
- lysozyme at 2 μL / mL of B-Per bacterial extraction reagent
- DNAse I at 2 μL / mL of B-Per bacterial extraction reagent
- AEBSF to a final concentration of 1 mM
- Solubilize the cell pellet in lysis buffer and vortex to mix.
- Incubate cell pellet in lysis buffer at room temperature for 15 min.
- To pellet the cell debris, centrifuge the lysate at 15,000 g for 30 min at 4 °C.
- Complete the next section (Prepare Ni-NTA affinity column) during the centrifugation.
Prepare Ni-NTA affinity column
- Obtain a 500 μL aliquot of 50% slurry (Ni-NTA resin) and mix the slurry by inverting the tube several times.
- The slurry is the Ni-NTA column matrix!
- Centrifuge the slurry for 30 sec then remove the supernatent.
- To wash the slurry, add 500 μL of 1X PBS and invert the tube 3 times.
- Add the slurry to the column and allow the 1X PBS to run through the column.
- Be sure a beaker is placed under the column to collect the waste!
- When the PBS has flowed through, cap the bottom and the top of the column until you are ready to add the cell lysate.
Purify PF3D7_1351100 from cell lysate
- Transfer the supernatent from the centrifuged cell lysate to a fresh microcentrifuge tube.
- Label the microcentrifuge tube containing the cell pellet as "pellet" and give it to the Instructor! This pellet will be used later when protein expression and purity are examined.
- Aliquot 30 μL of the supernatent (from Step #1) to a fresh microcentrifuge tube.
- Label the microcentrifuge tube containing the aliquot as "lysate" and give it to the Instructor! This aliquot will be used later when protein expression and purity are examined.
- Pipet the remaining supernatent into the prepared Ni-NTA affinity column.
- Be sure that the bottom of the column is capped!
- Attach the cap to the top of the column and incubate on the nutator for 2 hrs at 4 °C.
- Following the incubation, clamp the affinity column into the ring stand.
- Collect the flowthrough from the affinity column.
- Hold a microcentrifuge tube under the column, then remove the bottom cap from the column and collect the liquid that leaves the column.
- Label the microcentrifuge tube as "flowthrough" and give it to the Instructor! This aliquot will be used later when protein expression and purity are examined.
- To wash the Ni-NTA affinity column, add 10 mL of wash buffer.
- Hold a microcentrifuge tube under the column, then remove the bottom cap from the column and collect ~250 μL of the liquid that leaves the column.
- Label the microcentrifuge tube as "wash" and give it to the Instructor! This aliquot will be used later when protein expression and purity are examined.
- To elute the PF3D7_1351100 protein from the affinity column, add 1 mL of elution buffer.
- Hold a microcentrifuge tube under the column, then remove the bottom cap from the column and collect the entire 1 mL of the liquid that leaves the column.
- Label the microcentrifuge tube as "elution 1" and give it to the Instructor! This aliquot will be used later when protein expression and purity are examined.
- Repeat the elution procedure in Step #8 a total of three times.
- Label the microcentrifuge tubes as "elution 2" and "elution 3" then give to the Instructor! These aliquots will be used later when protein expression and purify are examined.
- Lastly, resuspend the slurry from the Ni-NTA affinity column in 250 μL 1X PBS and transfer to a fresh microcentrifuge tube.
- Label the microcentrifuge tube as "slurry" and give it to the Instructor! This aliquot will be used later when protein expression and purity are examined.
In your laboratory notebook, complete the following:
- At several steps in the protein purification procedure, samples are collected that will be used later when protein expression and purity are examined. Consider why each of the samples listed below are saved as controls to measure the success of the purification.
- The pellet from Step #1.
- The lysate from Step #2.
- The flowthrough from Step #6.
- The wash from Step #7.
- The slurry from Step #10.
- What is occurring during the incubation in Step #4?
Reagents list
- Terrific broth (TB) (from RPI)
- kanamycin (from Sigma)
- isopropyl β-d-1-thiogalactopyranoside (IPTG) (from Sigma)
- 2x B-Per bacterial protein extraction reagent (from ThermoFisher)
- lysozyme (from Sigma)
- DNase I (from Sigma)
- 4-(2-aminoethyl)benzenesulfonyl fluoride (AEBSF) (from Sigma)
- phosphate saline buffer (PBS) (from VWR)
- Ni-NTA agarose (from Qiagen)
- Wash buffer: 100 mM HEPES (pH = 7.4), 500 mM NaCl, 10 mM imidazole
- Elution buffer: 100 mM HEPES (pH = 7.4), 500 mM NaCl, 250 mM imidazole
- imidazole (from Sigma)
Next day: Assess purity and concentration of purified protein