Difference between revisions of "20.109(S08):Initiate cell culture (Day2)"
(→Cell culture) |
(→For next time) |
||
| Line 38: | Line 38: | ||
#The cells intended for your 2D culture probably do not need to be spun down again, but the cells for your 3D cultures must be directly resuspended in alginate rather than culture medium. While you spin down your alginate-bound cells, set up your 2D cultures according to the directions below. | #The cells intended for your 2D culture probably do not need to be spun down again, but the cells for your 3D cultures must be directly resuspended in alginate rather than culture medium. While you spin down your alginate-bound cells, set up your 2D cultures according to the directions below. | ||
#Label two T25 flasks with your group colour, initials, the date, and a description of the cells and culture conditions. Now make your cell dilution, using the example below as a guide - just change the volumes according to how many cells you want to add (always making 10 mL of culture total). | #Label two T25 flasks with your group colour, initials, the date, and a description of the cells and culture conditions. Now make your cell dilution, using the example below as a guide - just change the volumes according to how many cells you want to add (always making 10 mL of culture total). | ||
| − | #* | + | #*Let���s say you have 6M cells/mL in your conical tube, and want to use 600,000 cells to initiate your 2D cultures. Stand your T25 flasks upright and take the caps off. Start by adding 100 μL of chondrocytes against the back wall of each of your T25 flasks, making sure to touch only the sterile pipet tip to the flask, and to avoid touching the pipetman body inside this sterile area. (By back wall I mean what will become the bottom inside wall when you lay the flask down, which is the surface on which your cells grow.) |
#*Now add 9.9 mL of warm culture medium into each flask, and triterate to mix the cells. | #*Now add 9.9 mL of warm culture medium into each flask, and triterate to mix the cells. | ||
#*Finally, tighten the cap back onto the flask and put the cells in the incubator. | #*Finally, tighten the cap back onto the flask and put the cells in the incubator. | ||
| Line 58: | Line 58: | ||
Did you know that NCBI has a whole site devoted to [http://www.ncbi.nlm.nih.gov/projects/genome/guide/cow/ all things cow]? | Did you know that NCBI has a whole site devoted to [http://www.ncbi.nlm.nih.gov/projects/genome/guide/cow/ all things cow]? | ||
| − | + | It���s true! And today you will use this site to find the primers you need to perform RT-PCR on Day 4 of this module. Try searching for collagen types I and II (the alpha chain of each is fine) in the '''Map Viewer''' (upper right of page). What chromosomee is each collagen chain located on? See if you can make your way to the UniSTS entries for collagen, which list recommended primers for RT-PCR. How long are the expected PCR products if these primers are used? | |
Another option for finding primer suggestions is looking in the literature. Of course, this can be a risky proposition, but if you verify the primers against information in the NCBI database, it can be faster than making your own from scratch, and provide a feeling of security (someone, somewhere has succesfully amplified the sequence in question!). The paper by [ |Ikenooue et al.] lists primers recommended for collagen type II. What species are the primers for? If it's not bovine, you cannot use the primers directly. However, you can BLAST the primers against the bovine genome, similar to what you did in Module 2 to verify your mutagenized plasmids against the original. | Another option for finding primer suggestions is looking in the literature. Of course, this can be a risky proposition, but if you verify the primers against information in the NCBI database, it can be faster than making your own from scratch, and provide a feeling of security (someone, somewhere has succesfully amplified the sequence in question!). The paper by [ |Ikenooue et al.] lists primers recommended for collagen type II. What species are the primers for? If it's not bovine, you cannot use the primers directly. However, you can BLAST the primers against the bovine genome, similar to what you did in Module 2 to verify your mutagenized plasmids against the original. | ||
| − | Go to the [http://www.ncbi.nlm.nih.gov/blast/Blast.cgi BLAST site] and select the ''bos taurus'' genome. Type in the primers from the journal article one at a time, then perform the BLAST as follows: select BLASTN, change the | + | Go to the [http://www.ncbi.nlm.nih.gov/blast/Blast.cgi BLAST site] and select the ''bos taurus'' genome. Type in the primers from the journal article one at a time, then perform the BLAST as follows: select BLASTN, change the ���Expect��� value to 0.1, and turn off the low complexity filter. How many nucleotides changed between the human and cow for each primer? |
Why must you use cDNA rather than complete genes (introns+exons) when making primers for RT-PCR? | Why must you use cDNA rather than complete genes (introns+exons) when making primers for RT-PCR? | ||
| Line 68: | Line 68: | ||
==For next time== | ==For next time== | ||
| − | #[ | + | #Sign up for a time to do your Day 3 lab work on the Day 3 talk page; on this day you will arrive at staggered times. |
| − | #You should also continue choosing and thinking about the topic for your [ | + | #[http://openwetware.org/wiki/20.109(S08):Module_3_oral_presentations | The primary assignment] for this experimental module will be for you to develop a research proposal and present your idea to the class. For next time, please describe five recent findings that might define an interesting research question. You should hand in a 3-5 sentence description of each topic and list the reference that led you to each item. The topics you pick can be related to any aspect of the class, i.e. DNA, protein, or cell-biomaterial engineering. During lab next time, you and your partner will review the topics and narrow your choices, identifying one or perhaps two topics for further research. |
| + | #You should also continue choosing and thinking about the topic for your [http://openwetware.org/wiki/20.109(S08):Essay |essay]. You will be writing an essay draft about a week from now. Try to sketch a couple of supporting arguments that you might make for your position in a sentence or two. This part will not be collected. However, if you would like to write about something other than standards in the tissue engineering community, please let me know when you decide on a topic. | ||
==Reagent list== | ==Reagent list== | ||
Revision as of 20:03, 22 April 2008
Contents
Introduction
Last time you proposed culture conditions for an investigation of chondrocyte phenotype maintenance, and today you will initiate said cultures. The cells you are using are freshly derived from bovine cartilage: cells from cows are often used in part because of their availability from abattoirs. In general, large animals are more useful for modeling human joint diseases such as osteoarthritis than are small animals, because the resting angle of their knee joints is more similar to that of humans. In this module, we will work with an in vitro culture model of cartilage-forming cells.
Two of your three cell samples will be grown in alginate bead cultures. You have probably encountered alginates many times in your life, as thickeners in food and textiles, preservatives, and possibily at your dentist or in a pharmacy. Alginate is a polysacharride derived from seaweed, a co-polymer of mannuronic and guluronic acid. A single alginate molecule may contain long stretches of either acid (called M-blocks and G-blocks), as well as random and/or strictly alternating G/M sequences. The precise chemical composition of an alginate determines its mechanical properties, degradability, and other important characteristics. Qualities such as strength and viscosity are also influenced by the average length of the individual polymer chains (i.e., the molecular weight), and by alginate concentration. For example, high molecular weights correlate with increased viscosity. Alginates in general are shear-thinning, which is to say their viscosity decreases as shear rate increases (e.g., when quickly drawn into a syringe).
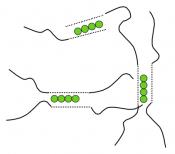
Cations such as calcium can cross-link alginate chains to form a network, or gel. The identity and concentration of the cross-linker influence the ultimate material properties. Only G-blocks can be linked to each other, while M- or MG-blocks cannot, but in turn provide flexibility (see figure). The resultant semi-solid structure has the capacity to hold a large amount of water, and the water-swollen structure is called a hydrogel. Hydrogels have several attractive properties for tissue engineering: they allow oxygen and nutrients to diffuse better than non-hydrated materials do; their mechanical and biochemical properties are readily varied by co-polymerization of multiple elements; they mimic the elasticity of natural tissues, and they often form rapidly and under mild conditions. Some gels can be injected into a patient in liquid form, then solidified within his or her body by heat or light. Such injectable gels have the advantage of easily filling an arbitrarily sized wound shape, which is difficult for implantable gels to do. Natural (e.g., alginate) and synthetic (e.g., poly(ethylene glycol)) hydrogels each have distinct advantages and disadvantages, as we will discuss in class.
Today you will make alginate hydrogels in bead form, by slowly releasing alginate solution from a syringe into a bath containing calcium chloride. Next time you will see how well your cells survive in both alginate and monolayer culture.
Protocols
Half the class at a time will work in the tissue culture room today. The other half of you will explore the NCBI bovine information site, and otherwise spend the time however you find useful (FNT assignment, notebook prep, or unrelated work).
Part 1: Chondrocyte cell culture
Today you will work with primary cells that are directly isolated from bovine knee joints. Yesterday, your teaching faculty harvested cartilage fragments from two bovine knees, and sequentially digested them in pronase and collagenase enzymes. Each joint typically yields > 100M cells, which should be plenty for half the class to work with. For the first part of your time in TC, you will observe how we get from a digested tissue product to a full cell isolate. Then you will each plate out your 2D and 3D cultures.
Cell isolation
During the centrifugation steps, you can work on setting up your hoods (they have been partially set up for you, but you can start labeling your tubes and flasks now) and preparing your lab notebooks. We will also discuss each of your experimental plans.
- The digested cell solution is filtered into 2 tubes using a cell strainer with a 70 μm cutoff size. This is to remove undigested debris.
- A second filtration is done at a 40 μm cutoff size, to remove finer debris. (What else might be removed at this stage, for better or for worse?)
- The cells are centrifuged for 6 min at 1900 g.
- The enzymatic solution is aspirated, and the cells are initially resuspended in a 1 mL volume of sterile PBS, then combined in one tube at 25 mL total. (What would happen if you tried to resuspend the cells in a large volume right away?)
- The cells are spun down and resuspended in sterile PBS again.
- Finally, they are spun down and resuspended in pre-warmed culture medium, 20 mL total.
- We will now count the cells, diluting 50 μL in 200 μL PBS, then mixing 1:1 with Trypan blue. Each group will be given one-third of the cells.
Cell culture
- Look over the plan you made last time: do you have enough cells to make the cultures that you planned? If not, make a revision to your experiment; otherwise, proceed.
- Separate the cells that will make up your three different cultures into three labeled 15 mL conical tubes. Note that the tubes may not all require the same amount of cells, depending on the cell densities you chose for the three cultures. Double-checking your calculations now may save you having to do an extra centrifugation step later!
- The cells intended for your 2D culture probably do not need to be spun down again, but the cells for your 3D cultures must be directly resuspended in alginate rather than culture medium. While you spin down your alginate-bound cells, set up your 2D cultures according to the directions below.
- Label two T25 flasks with your group colour, initials, the date, and a description of the cells and culture conditions. Now make your cell dilution, using the example below as a guide - just change the volumes according to how many cells you want to add (always making 10 mL of culture total).
- Let���s say you have 6M cells/mL in your conical tube, and want to use 600,000 cells to initiate your 2D cultures. Stand your T25 flasks upright and take the caps off. Start by adding 100 μL of chondrocytes against the back wall of each of your T25 flasks, making sure to touch only the sterile pipet tip to the flask, and to avoid touching the pipetman body inside this sterile area. (By back wall I mean what will become the bottom inside wall when you lay the flask down, which is the surface on which your cells grow.)
- Now add 9.9 mL of warm culture medium into each flask, and triterate to mix the cells.
- Finally, tighten the cap back onto the flask and put the cells in the incubator.
- Now you are ready to initiate the alginate cultures. Label two six well plates with the same information as for your 2D cultures, using one plate per culture type (e.g., plate 1 = low cell density, plate 2 = high cell density).
- Resuspend your cells in the appropriate amount of the type and concentration of alginate that you chose.
- Using the syringe that has been prepared for you, very carefully pull up the cells, then release them drop-by-drop into the beaker full of calcium chloride solution (20 mL). Recall that calcium effectively polymerizes the alginate, resulting in small gel beads filled with cells. Immediately discard the entire syringe into the sharps container - do not try to remove or recap the needle.
- Depending on the concentration of alginate that you chose, you may have between ~50-150 beads for 1 mL of alginate solution.
- Allow the polymerization to proceed for 10 min. at room temperature. Then pour your beads into a 50mL conical tube.
- Remove the calcium chloride solution from your beads using a large serological pipet (to better avoid aspirating the beads), and put this solution in the temporary waste beaker in your hood.
- Now fill the conical tube with sodium chloride (20 mL), and gently shake it for 1-2 min. This is to remove excess calcium from the solution.
- Remove the NaCl using a fresh pipet, then wash the beads again with fresh NaCl. Finally, wash the beads two times with DMEM culture medium, first with the additive-poor and then with the additive-rich DMEM (20 mL each time).
- For each of your two samples, transfer the beads to the two leftmost wells of a 6-well plate, using a sterile spatula. Try to put approximately equal numbers of beads in the two wells.
- Finally, add 6 mL of warm culture medium to your four sample wells, then put the two well-plates in the incubator.
The teaching faculty will exchange the culture medium and split the cells as necessary.
Part 2: Primers for RT-PCR
Did you know that NCBI has a whole site devoted to all things cow?
It���s true! And today you will use this site to find the primers you need to perform RT-PCR on Day 4 of this module. Try searching for collagen types I and II (the alpha chain of each is fine) in the Map Viewer (upper right of page). What chromosomee is each collagen chain located on? See if you can make your way to the UniSTS entries for collagen, which list recommended primers for RT-PCR. How long are the expected PCR products if these primers are used?
Another option for finding primer suggestions is looking in the literature. Of course, this can be a risky proposition, but if you verify the primers against information in the NCBI database, it can be faster than making your own from scratch, and provide a feeling of security (someone, somewhere has succesfully amplified the sequence in question!). The paper by [ |Ikenooue et al.] lists primers recommended for collagen type II. What species are the primers for? If it's not bovine, you cannot use the primers directly. However, you can BLAST the primers against the bovine genome, similar to what you did in Module 2 to verify your mutagenized plasmids against the original.
Go to the BLAST site and select the bos taurus genome. Type in the primers from the journal article one at a time, then perform the BLAST as follows: select BLASTN, change the ���Expect��� value to 0.1, and turn off the low complexity filter. How many nucleotides changed between the human and cow for each primer?
Why must you use cDNA rather than complete genes (introns+exons) when making primers for RT-PCR?
For next time
- Sign up for a time to do your Day 3 lab work on the Day 3 talk page; on this day you will arrive at staggered times.
- | The primary assignment for this experimental module will be for you to develop a research proposal and present your idea to the class. For next time, please describe five recent findings that might define an interesting research question. You should hand in a 3-5 sentence description of each topic and list the reference that led you to each item. The topics you pick can be related to any aspect of the class, i.e. DNA, protein, or cell-biomaterial engineering. During lab next time, you and your partner will review the topics and narrow your choices, identifying one or perhaps two topics for further research.
- You should also continue choosing and thinking about the topic for your |essay. You will be writing an essay draft about a week from now. Try to sketch a couple of supporting arguments that you might make for your position in a sentence or two. This part will not be collected. However, if you would like to write about something other than standards in the tissue engineering community, please let me know when you decide on a topic.